- TrumpRx to expand coverage to more than 800 drugs
- FDA approves new treatment for children with hemophilia B
- ‘Smart growth’ is a necessity for Missouri system now serving 4 states: CEO
- 10 most-viewed psychiatry residency programs in 2025
- 10 providers seeking RCM talent
- NYC Health + Hospitals faces Senate scrutiny after cyber incident
- Stamford Health plans $275M cancer, behavioral health expansion
- 7 dentists making headlines
- 49 top-rated RCM vendors for 2026, per Black Book
- 10 AI vendors gaining traction with health systems
- ‘Find your purpose’: Ascension CFO’s advice to finance leaders
- Measles cases pass 2,000, approach 2025 total: 4 notes
- Former clinic CEO pleads guilty to filing false tax returns
- Cleveland Clinic taps new chair of general surgery
- North Carolina heart tower project enters CON race
- Hartford Healthcare acquires cardiology group after antitrust settlement
- Just 5 Minutes Of Prayer Helps Reduce Pain and Anxiety, Study Finds
- Medtronic Advances Hugo Robotic Surgery Platform with Key FDA Filings and Product Approvals
- Medtronic Posts Strongest Revenue Growth in a Decade, Driven by Cardiovascular and Surgical Businesses
- ADA: Novo's Wegovy pill reaches new GLP-1 patients with impressive 3M prescription milestone
- Boston Scientific Plans Indiana Distribution Center, 300 New Jobs
- ADHD ‘Masking’ May Help People Blend In But Harms Mental Health
- Why Alcohol Makes You Crave Salty Snacks — And How Protein-Rich Foods Can Help Prevent Weight Gain
- Getting The RSV Shot While Pregnant Could Protect Your Baby After Birth
- Irregular Sleep Risks Preschool Kids' Brain Power
- Pfizer scores FDA pediatric expansion for hemophilia treatment Hympavzi
- Upcoming Billing Change Could Make Pregnancy Pricier
- MAHA’s Treatments for Autism: Camel’s Milk, Stem Cell Injections — And Spelling Therapy
- By September, Nearly a Third of Americans Will Live in States With Legal Aid in Dying
- Hikma earmarks $267M to expand pair of Ohio production sites, add 350 jobs
- Stepful banks $55M to scale AI-powered medical training to boost the workforce pipeline
- ADA: Amgen highlights Repatha perks, GLP-1 pitfalls in push to boost cardiometabolic pedigree
- Gounder Fills In Details Behind Ebola, GLP-1, and Trump Headlines
- Untreated Cancer, Festering Infections: Immigrant Detainees Detail Medical Care Lapses
- Elation Health acquires Aster as it ramps up agentic AI for primary care
- Virginia dental practice suffers data breach affecting 5,200 individuals
- CRNAs could gain foothold in Medicare payment models
- Virginia oral surgery practice suffers data breach affecting 7,000 individuals
- 3 cases redrawing the lines of physician contracting
- ‘IDR is the exception, not the rule’: TeamHealth CMO on billing reality
- States ranked by mental health social workers per capita
- UnitedHealthcare cut CRNA pay 15% — are ASCs picking up the tab?
- Waco Cardiology’s research arm acquired in 3-city deal
- New York opens 60-bed OUD recovery center
- Kratom admissions jump 525% at California treatment center
- Buy, join or partner? Dentists’ advice for new grads
- The 10 states where ophthalmologists’ pay climbed the most
- Addiction Recovery Care founder indicted on fraud, money laundering charges
- Dengue Is No Longer Just A Travel Risk — What Google’s Mosquito Plan Could Mean For Your Summer
- Ohio targets behavioral health fraud with new oversight measures
- 12 state behavioral health policy updates
- California dental practice adds humanoid robot for front-desk help
- DentaQuest cybersecurity incident exposes 2.6 million accounts
- The 5 states with the most DSO action in May
- 5 highest-paying cities for dental assistants in 2026
- 5 highest-paying cities for hygienists in 2026
- 3 dental acquisitions in May
- Brain Surgery For Pituitary Tumor Helps Illinois Mom Have Second Baby
- Essence Healthcare, Oura expand partnership to sleep apnea detection
- Takeda takes $2.5B legal charge after pay-for-delay verdict, swings to loss for '25
- Industry Voices—A healthcare experience that is as seamless as it is soulful
- Fullspan Health debuts Healthline AI companion to connect consumers to medical content and providers
- As pro-life pressure ramps up on Trump, FDA is investigating safety of abortion pill: WSJ
- As pro-life pressure ramps up on Trump, FDA is investigating safety of abortion pill: WSJ
- Popular Blood Pressure Meds, Dihydropyridine Calcium-Channel Blockers, Linked To Kidney Damage Risk In Type 2 Diabetes
- Too Much Sitting In Pregnancy Doubles Risk Of Complications
- Spinal Cord Stimulation May Restore Arm Strength After Stroke
- Behind the headlines of ASCO 2026
- RFK Jr. Seeks To Peek At Americans' Medical Records For Clues On Autism And Vaccines
- Two-Pill Combo, Inqovi and Venclexta, Offers Hospital-Free Leukemia Treatment Option For Older Adults With AML
- ‘We Live With Fear’: In Congo, Doctors Face Ebola With Little Protection
- Millions of Kids Could Lose Insurance as GOP Healthcare Cuts Start To Bite
- Upcoming Billing Change Could Make Pregnancy Pricier
- WellSpan Health strikes strategic partnership with Philips to deploy, co-develop new healthcare tech
- Oregon overhauls 30-year behavioral health agreement model: 3 things to know
- 6 states regulating AI in mental health
- RWJBarnabas Health sets 15K target for naloxone kit program
- At first public hearing for FDA’s CNPV program, industry support meets ‘deep concern’ from experts
- “Harmonization: We’ll Have Lots to Talk About”
- Remarks at the Investor Advisory Committee Meeting
- A Quarter for your Thoughts: Remarks at the Meeting of the SEC Investor Advisory Committee
- Weekly Rundown: Trump signs AI executive order; Health Catalyst to sell Vitalware for $147M
- Small businesses feel the squeeze as healthcare costs rise: Morgan Health
- Poll Finds Broad Support For Stricter Regulations On Ultra-Processed Foods
- It's unanimous: SCOTUS agrees with Hikma in 'skinny label' case vs. Amarin
- Remarks at the Investor Advisory Committee Meeting
- Fierce Pharma Asia—China's biotech rise; ASCO highlights; Lilly pipeline deals
- Nearly 1 in 5 Young People Turn to AI Chatbots for Mental Health Advice
- Ixlayer rolls out digital acute pain management platform in collaboration with Vertex Pharmaceuticals
- Georgetown study: 2M children have dropped out of Medicaid, CHIP since January 2025
- ScionHealth sends 8 community hospitals to Lifepoint Health
- ADC Therapeutics' shares plummet after patient deaths in trial of Zynlonta
- As China biotech crackdown calls reverberate in Washington, the pushback gets louder, too
- Older LGBTQ+ Adults Fear Less Support As They Age, Poll Shows
- Night Owls Are More Prone To Anxiety, Loneliness
- Home-Delivered Medical Meals Reduce ER Visits, Save Money
- Axsome fends off generic competition to narcolepsy med Sunosi until 2040
- Menopause Hormone Therapy Use Drops Sharply Across United States
- Michigan Found A Way To Reduce School Vaccine Waivers — Until It Backfired
- Listen to the Latest ‘KFF Health News Minute’
- Louisiana’s Reporting Law Chills Immigrant Medicaid Applications
- RFK Jr. Seeks To Peek at Americans’ Medical Records for Clues on Autism and Vaccines
- Thyme Care expands cancer survivorship program to provide longitudinal support
- Medline earns FDA warning letter ire for repeated toxic bacteria problems
- 8 Requirements for AI Medical Interpreting: 2026 Survey Results
- Commonwealth Fund: 21% of adults experienced a coverage denial in the past year
- Healthcare Dealmakers—WakeMed to join Atrium Health, Qualtrics closes $6.8B Press Ganey deal and more
- Millions on Medicaid May Soon Have To Prove They’re Working To Keep Coverage
- Anomaly Insights launches AI solution for managed care executives
- Lilly, Boehringer to slash at least $1B each from planned investments in Germany
- Presbyterian Healthcare Services to discontinue MA plans in 2027, cut 150 jobs
- Just 90 Minutes Of Strength Training A Week Linked To Longer Life
- Amid Miplyffa launch, Zevra CEO aims to foster EU-style Niemann-Pick market in US
- Chemo-Free Drug, Tecvayli, Shows Major Survival, Remission Gains In Relapsed Multiple Myeloma
- Urine Test Can Detect Autism, Study Says
- High-Puff Vapes Become More Toxic Over Time, Study Says
- With Cencora pact, Gilead looks to grow CAR-T treatment center network
- Coffee, racetracks, beaches and more coffee—inside the ASCO 2026 exhibit floor
- GLP-1 Drugs Like Ozempic May Lower Breast Cancer Risk By About 30%
- Telehealth Booms As Demand For GLP-1s Surges and Questions Mount About Safety, Oversight
- A patient-first shift in disease language and communication
- FDA issues untitled letter after finding ‘eye-catching graphics’ in promotional emails
- Neurotech launches new YouTube channel to spotlight patients’ stories for rare eye disorder
- As Akeso takes center stage at ASCO, China biotech industry cements its coming of age
- Base Case: Remarks at the IC3 Blockchain Camp
- Department of Labor's proposed foreign worker wage increases are more tough news for healthcare staffing
- Trump Signs Order Calling For Fewer Childhood Vaccines
- Ascension closes its $3.9B AmSurg purchase following FTC's all-clear
- Simple Blood Test May Help Detect And Stage Alzheimer's Disease
- Smartphone App Helps Those With Advanced Cancer Maintain Quality Of Life
- Asthma Drug, Tezspire, Cuts Need For Steroid Pills While Keeping Attacks In Check
- Childhood Flu Shots Prevent Millions of Cases, Study Finds
- Amid Ebola, Hantavirus Outbreaks, Democrats Decry Trump's Health Cuts
- TikTok Videos Fuel Illicit Vaping Culture Among Underage Youth
- Commission Statement on the Passing of Former General Counsel David Becker
- Your Surprise Medical Bill May Be Gone — But Your Premiums Could Still Spike
- Short-Term Fasting Could Boost Chemo Response in Ovarian Cancer, Study Suggests
- MedTech In Focus: AI impact in healthcare
- If Your AI Can’t Explain Itself, Can FDA Authorize It?
- Workout Habits May Protect Against Inherited Heart Problems
- Childhood Lying Is Normal and Rarely Signals Behavioral Concerns, Study Says
- Perfectionism Among College Students Reaches Record High, Fueling Anxiety
- After Her Bout of Amnesia, A $59,000 Billing Dispute Wouldn't Go Away
- Rising Stars: WPP’s Meghan O’Hora on the ‘complex puzzle’ of oncology drug marketing
- Climate Change: Statement on Proposed Rescission of Climate-Related Disclosure Rules
- Statement of Commissioner Mark T. Uyeda on the Rescission of Climate-Related Disclosure Rules
- Keynote Remarks at the 2026 Reagan National Economic Forum
- Statement on Proposing Release for Rescission of Climate-Related Disclosure Rules
- Privacy and PetShops: Remarks at the Regulatory PETshop Series: Cryptographic Technologies and Financial Services Regulation
- CAT on a Hot Tin Roof
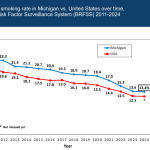
Michigan healthcare freedom community forum
Denmark may seem to be an odd exemplar for health care policy, but they are home to Novo Nordisk A/S, the worlds largest and most sophisticated manufacturer of pharmaceutical products and provider of health care services. The revolving door of the Indenrigs-og Sundhedsministeriet (Danish Ministry of Health) is connected to Novo Nordisk:
https://thehill.com/policy/healthcare/5656844-trump-rfk-child-immunization-schedule-overhaul/
HHS to announce overhaul to child vaccine schedule, aligning with Denmark
By Joseph Choi - December 19, 2025The Trump administration is planning to announce an overhaul to the childhood immunization schedule early next year, recommending fewer shots and aligning U.S. policy with that of Denmark.
A source familiar with the matter told NewsNation correspondent Libbey Dean that a new vaccination schedule to be released next year would recommend fewer shots, “aligning more with Denmark’s vaccination schedule.”
The Department of Health and Human Services (HHS) was scheduled to make an announcement on “children’s health” Friday, but it was postponed because of competing plans from the White House, which is expected to announce new deals to lower drug prices Friday afternoon.
HHS Secretary Robert F. Kennedy Jr. and his allies have routinely cited Denmark as an ideal in terms of vaccine policy that the U.S. should emulate. Denmark’s immunization schedule does not recommend seasonal vaccines for respiratory viruses like RSV, COVID-19 and the flu. The country also doesn’t recommend vaccinating against hepatitis B for all infants or against chickenpox.
During a recent meeting of the Advisory Committee on Immunization Practices (ACIP) for the Centers for Disease Control and Prevention (CDC), members of the panel sympathetic to Kennedy repeatedly pointed to Denmark’s immunization policy, with a presentation on the differences between American and Danish vaccine schedules included in the agenda.
The presentation noted the U.S. recommends 72 childhood vaccination doses targeting 18 diseases, while Denmark only recommends 11 doses targeting 10 diseases.
Pro-vaccine voices critical of Kennedy’s frequent comparisons to Denmark argue that the small European nation does not make for a fair comparison. The country has a population of roughly 6 million people distributed across an area similar to the state of Maryland. Denmark also has universal health care system with free comprehensive health services.
Maybe not. Big Pharma has weighed in:
https://www.politico.com/news/2025/12/20/rfk-kennedy-danish-vaccine-schedule-denmark-00701999
RFK Jr. wanted to endorse the Danish vaccine schedule. He was forced to pull back.
Legal and political concerns prompted the health department to cancel a planned announcement on Friday, officials said.
By Tim Röhn - December 20, 2025Health Secretary Robert F. Kennedy Jr. came within hours of publicly promoting Denmark’s childhood vaccine schedule as an option for American parents — before legal and political concerns got in the way.
A senior HHS official told POLITICO that a press conference set for Friday was canceled at the last minute after the HHS Office of the General Counsel said it would invite a lawsuit the administration could lose.
A second senior official at the Department of Health and Human Services confirmed the press conference, which HHS had publicly announced, was to be about the Danish schedule. The second official said it was canceled because it was deemed politically risky.
Billed as an “announcement regarding children’s health,” Kennedy was to appear alongside his top agency heads and Tracy Beth Høeg, the Food and Drug Administration’s top drug regulator. Høeg touted the Danish schedule at a vaccine advisory committee meeting earlier this month.
HHS canceled the event Thursday evening, hours after announcing it.
Andrew Nixon, an HHS spokesperson, called accounts of the cancellation that didn’t come directly from the department “pure speculation” in a statement.
HHS officials skeptical of moving to the Danish schedule, which recommends immunization for only 10 of the 17 diseases on the U.S. list, were relieved it was never publicly recommended, the first official said. The internal confusion and disagreement follow similar management bungling within HHS’ Food and Drug Administration that has frustrated the White House.
On Dec. 5, President Donald Trump signed a presidential memorandum titled “Aligning United States Core Childhood Vaccine Recommendations with Best Practices from Peer, Developed Countries.” The memorandum directed HHS and the Centers for Disease Control and Prevention, one of its subagencies that sets the vaccine schedule, to review peer-country best practices for vaccines recommended for all children and, if those practices were judged superior, to update the U.S. schedule while preserving access to vaccines already available. In the memorandum, Trump mentioned Denmark, Japan and Germany as examples of countries that recommend fewer shots than the U.S.
According to the first official, Kennedy and his top aide, Stefanie Spear, helped sell the peer-country framing to West Wing officials as the clearest way to turn internal vaccine skepticism into a signed White House directive. Spear is Kennedy’s principal deputy chief of staff and senior counselor.
Kennedy is a longtime vaccine skeptic who believes the U.S. schedule has grown too quickly, has not been tested in its entirety for adverse effects, and is a likely cause of rising autism rates. Numerous studies have not found a link between vaccines and the neurological disorder that now affects one in 31 U.S. children, up from one in 150 two decades ago.
Experts in the condition, which affects the ability to communicate, say expanded diagnostic criteria and awareness are responsible for most of that rise. The condition’s cause is usually genetic, they believe, but researchers are studying possible environmental causes.
HHS has made it a priority to learn more about what causes autism and why diagnoses are rising. The department’s research arm, the National Institutes of Health, announced an Autism Data Science Initiative on May 27 and has awarded around $50 million to fund 13 projects investigating potential causes.
In April, Kennedy promised to reveal autism’s cause in September, but HHS later said it would reveal preliminary findings early next year. Autism researchers, who have studied the condition for years, have called that unrealistic.
The first indication Kennedy might be considering the slimmer Danish schedule, which excludes vaccines for chickenpox, the flu, hepatitis A and B, meningitis, respiratory syncytial virus and rotavirus, came earlier this month during the CDC’s Advisory Committee on Immunization Practices meeting in Atlanta. Høeg presented a slide deck titled “U.S. vs. Danish Vaccine Schedule,” which the CDC posted among the meeting presentations.
The department then circulated Høeg’s presentation to top officials at HHS, the first senior official said. In the ensuing debate, Høeg’s supporters proposed offering the Danish schedule as a government-recommended alternative to the U.S. one.
The first senior official and two others inside HHS familiar with internal discussions, all of whom were granted anonymity to reveal deliberations they were not authorized to discuss publicly, said proponents of the Danish schedule felt that offering it would help restore trust in vaccines; many Americans were turned off by Covid-era vaccine mandates and claims that Covid shots would halt transmission that turned out to be incorrect, they argued.
The three officials said the view of proponents inside the administration was that the Danish schedule could be pitched as a “reset” that might convince hesitant parents to vaccinate their kids.
Critics inside the administration, the officials said, argued the plan to recommend the Danish schedule was not rigorous and science-based — and that promoting it publicly would invite criticism. Rather than restoring trust, they said it could undermine it by signaling doubt about the need for, and safety of, routine immunization.
Going forward without laying the scientific groundwork or going through normal regulatory processes could also make the department vulnerable to lawsuits, the HHS general counsel’s office argued, according to the first senior official. Mike Stuart, who was a U.S. attorney in West Virginia in Trump’s first term, now is HHS general counsel.
The American Academy of Pediatrics, which represents doctors who care for children, along with other physician and public health groups, has already sued HHS for changes it made earlier this year to Covid vaccine recommendations, saying the department violated rules governing how regulatory changes are made. That case is pending in federal district court in Boston.
HHS has stopped recommending Covid boosters for previously vaccinated people under 65 who are not at high risk of the disease. Instead, the department says Americans should talk to their doctor and make a shared decision.
"Science Based" has become shorthand for making someone a lot of money.
HHS' new, Danish inspired, childhood vaccine schedule was announced today:
HHS dramatically overhauls childhood vaccine schedule, downgrading advice for flu, meningitis, other shots
Federal officials say the U.S. schedule should be more in line with recommendations in other countries. Public health experts say the goal belies differences between health systems and disease prevalence.
By Lauren Gardner | January 5, 2026The Department of Health and Human Services announced Monday a dramatic overhaul of the childhood vaccine schedule, weakening recommendations for flu and three other shots in the name of aligning U.S. policies with certain European countries.
The shift, which was telegraphed weeks ago before being paused amid legal and political concerns, comes after President Donald Trump directed Health Secretary Robert F. Kennedy Jr. in December to review the childhood schedule and consider revising it to align with those of other developed countries, most of which recommend fewer shots.
Kennedy, a longtime anti-vaccine activist before entering government, has criticized the American schedule for years as bloated compared to other nations. He has claimed associations between the schedule’s expansion since the late 1980s and growing rates of chronic conditions from autism to allergies — despite scientific consensus rejecting such links.
The Centers for Disease Control and Prevention will soften its advice for vaccines targeting rotavirus, meningococcal disease, hepatitis A and influenza, which the agency reported Monday has already killed nine American children so far this winter season.
Those shots will no longer be universally recommended but advised under a concept known as shared clinical decision-making, which emphasizes conversations between health care providers and patients before choosing to vaccinate. The agency shifted to such an approach last year for Covid-19 and hepatitis B vaccine recommendations for most populations.
The respiratory syncytial virus antibody injection for infants will be recommended for “certain high-risk groups or populations,” HHS said, but it’s unclear how officials are defining that group.
“At a time when parents, pediatricians and the public are looking for clear guidance and accurate information, this ill-considered decision will sow further chaos and confusion and erode confidence in immunizations,” American Academy of Pediatrics President Andrew Racine said in a statement. “This is no way to make our country healthier.”
The CDC also changed its recommendation Monday for the HPV vaccine to a single dose, despite FDA approval for a two- and three-dose series depending on a patient’s age.
The CDC will continue to recommend that kids be vaccinated against measles, mumps, rubella, polio, whooping cough, tetanus, diphtheria and human papillomavirus or HPV, diseases for which HHS said there’s “international consensus” in favor of immunization. The agency will also maintain its recommendation for the chickenpox vaccine, which some countries don’t embrace but others are increasingly advising.
With Monday’s changes, the core U.S. immunization schedule has been whittled down by about a third, to 11 shots from 18 in 2024. HHS Deputy Secretary Jim O’Neill, who’s currently acting as the CDC’s leader, signed a memo Monday directing the agency to make the changes effective immediately.
In doing so, the agency circumvented its own external advisory panel, the Advisory Committee on Immunization Practices, which has historically vetted schedule changes as new products are approved and safety and effectiveness data emerge. HHS also did not provide evidence in support of its decisions outside of an assessment conducted by two agency officials who have promoted European nations’ approaches to vaccines.
The presidential memo cited Denmark, Germany and Japan as “peer, developed” nations with slimmer schedules. Denmark’s schedule, which has been a chief point of comparison for HHS officials, does not recommend any of the shots that are being moved to shared clinical decisionmaking.
“The U.S. is a global outlier among peer nations in the number of target diseases included in its childhood vaccination schedule and in the total number of recommended vaccine doses,” Tracy Beth Hoeg, the FDA’s acting director for the Center for Drug Evaluation and Research, and Martin Kulldorff, chief science and data officer for HHS’ Office of the Assistant Secretary for Planning and Evaluation, wrote in an assessment upon which the department is basing its changes.
The assessment found that public trust in routine pediatric vaccines plummeted in the U.S. amid pandemic-era policies like school closures and immunization mandates in some areas of the country. It calls for softening some longstanding recommendations for illnesses that Hoeg and Kulldorff suggested aren’t as serious or widespread in a bid to restore public trust.
“The backdrop for this then meant that we needed to take action to increase the public confidence in the vaccine schedules,” a senior HHS official told reporters on a call Monday featuring several department officials, all of whom declined to comment on the record.
But vaccine and public health experts have said such a move belies fundamental differences between the fragmented U.S. health system and other countries — especially those in Europe — that provide universal care to their residents. Disease prevalence also varies across countries, and those experts say the longstanding U.S. recommendations reflect pathogens that commonly circulate here.
Many of the decisions other countries “make about vaccine schedules are not based on the burden of disease, but rather on cost,” Sean O’Leary, chair of the American Academy of Pediatrics’ committee on infectious diseases, told reporters.
The CDC’s vaccine advice has traditionally fallen into the same nuanced categories as those outlined Monday by Kennedy’s HHS: recommendations for all, recommendations for certain populations or high-risk groups, and individualized recommendations based on conversations between providers and patients.
A “recommended” vaccine means vaccination is the default, though the federal government doesn’t mandate vaccines for children to enter childcare facilities or schools. That power is left to the states.
Historically, the CDC only amended its immunization schedules after receiving input from the ACIP, which also has exclusive authority to direct changes to a federal safety-net program offering free vaccines to un- and underinsured children. Kennedy universally overhauled the panel last year to include several members who share his vaccine skepticism; the committee had already acted to weaken recommendations for Covid and hepatitis B vaccines before Monday’s announcement.
The senior HHS official said Monday’s changes don’t mean the advisory panel is being disbanded even though the department has circumvented its role in announcing the new changes.
“We will continue to pose important questions for them,” he said. “We will continue to update the schedule based on their advice, their thorough examination of particular vaccines, two or three at a time.”
HHS officials said all vaccines the CDC had recommended as of the end of 2025 will still be fully covered by federal insurers like Medicaid and by Affordable Care Act insurance plans. “This means that insurance will continue to cover more vaccines for children in the U.S. than in peer nations, where insurance generally only pays for recommended vaccines,” the department said in a fact sheet.
The AAP sued Kennedy last summer over the ACIP overhaul. O’Leary said he couldn’t comment on whether the group would amend the lawsuit to include Monday’s action.
O’Leary said Kennedy “does have a lot of leeway” to influence vaccine recommendations. But he and other public health experts said lawmakers should consider whether HHS is subverting congressional intent by making fundamental vaccine policy changes outside the traditional structure.
“The power to prevent the downstream consequences, that may end up being up to Congress,” he said.
Sen. Bill Cassidy (R-La.), who leads the chamber’s health policy committee and who cast the deciding vote last year recommending Kennedy’s confirmation, posted on X Monday that the CDC vaccine schedule “IS NOT A MANDATE,” though he didn’t call out the secretary by name.
“It’s a recommendation giving parents the power,” he said in his post. “Changing the pediatric vaccine schedule based on no scientific input on safety risks and little transparency will cause unnecessary fear for patients and doctors, and will make America sicker.”
The immediate public reaction of high profile Michigan health care providers is derogatory:
Michigan doctors criticize changes to US childhood vaccination schedule
By Eli Newman - January 5, 2026
- The CDC is scaling back the number of shots the public agency recommends for American children, moving to 11 from 17
- Doctors and public health experts in Michigan say the decision is politically motivated and will sow ‘chaos’ among patients and their families
- The Michigan Department of Health and Human Services issued an order last year to follow the immunization guidance laid out by the country’s leading physician groups
Some Michigan physicians and public health experts say the federal government’s decision to scale back its recommendations for childhood vaccinations could be dangerous and sow “chaos” among families and their doctors.
The US Centers for Disease Control and Prevention announced Monday it would update the immunization schedule for American youth by reducing the number of recommended routine shots to 11 from 17. The decision is tied to President Donald Trump’s order last year to review vaccine policy in other countries and update the American recommendations accordingly.
“After an exhaustive review of the evidence, we are aligning the US childhood vaccine schedule with international consensus while strengthening transparency and informed consent,” Robert F. Kennedy Jr., secretary of US Health and Human Services, said in a statement announcing the changes. “This decision protects children, respects families, and rebuilds trust in public health.”
But several Michigan doctors say the decision does the exact opposite.
“It will create confusion in the minds of parents and the American public,” said Dr. Anand Parekh, chief health policy officer at the University of Michigan School of Public Health.
A former US Health and Human Services deputy assistant secretary, Parekh said the CDC’s decision is “not based on science” and reflects the preferences of individual policymakers at the helm of the government. He said the change could be “quite harmful” in dissuading families from vaccination against preventable illnesses.
Following the announcement, the Michigan Department of Health and Human Services reiterated its support of vaccines as a successful tool to prevent the spread of infectious diseases, pointing to decisions made last year to maintain their use in the state among a shifting stance from federal policymakers.
In December, Michigan’s chief medical executive, Dr. Natasha Bagdasarian, issued a standing recommendation to follow the childhood immunization standards provided by the country’s leading physician groups, saying at the time that many public health experts “are not in agreement with recent federal vaccine recommendations.”
The American Academy of Pediatrics and American Academy of Family Physicians have criticized the immunization policy changes undertaken during the Trump administration and maintain childhood vaccination schedules that recommend more shots than the federal government.
Dr. Joseph Fakhoury, president of the Michigan chapter of the American Academy of Pediatrics, said the federal government’s decision would bring about “chaos” for families.
“It’s, truthfully, incredibly disappointing,” Fakhoury told Bridge. “Even those that already have a pretty strong confidence in immunizations and a good trusting relationship with their physician team … I worry more that it just will continue to erode that confidence.”
Vaccines for measles, mumps, rubella, polio, pertussis, tetanus, diphtheria, Haemophilus influenzae type B, pneumococcal disease, human papillomavirus and chickenpox will remain recommended for children under the updated federal policy. Shots to prevent influenza, hepatitis A and B, rotavirus, meningococcal disease and COVID-19 have shifted to a “shared decision-making” category, in which the government recommends consultation with a health care provider.
“All vaccines currently recommended by CDC will remain covered by insurance without cost sharing,” Centers for Medicare and Medicaid Services Administrator Dr. Mehmet Oz said in a statement. “No family will lose access. This framework empowers parents and physicians to make individualized decisions based on risk, while maintaining strong protection against serious disease.”
Those representing many of Michigan’s health insurance companies say they are not aware of any plans to change coverage.
“Customers’ access to essential vaccinations of their choice, as well as other forms of preventive care, is crucial to ensuring affordable health care and positive health outcomes,” said Brian Mills, spokesperson for the Michigan Association of Health Plans. “If customers have any questions about their current vaccination coverage, they’re encouraged to contact their health plan.”
Some vaccine proponents had worried the CDC would stop recommending the measles, mumps and rubella (MMR) vaccine after some, including President Donald Trump, have tried to link the shot to autism in children. While the American Psychiatric Association and other major physician groups discredit the claim, the CDC’s website now says the link can’t be ruled out.
Recent polling suggests 1 in 6 parents are skipping or delaying shots for their children, with skepticism toward vaccine efficacy and safety more prevalent among the GOP. According to Pew Research, 52% of Republicans support the school mandate for the MMR vaccine.
Oh, but it's the people who are confused!
https://www.michigan.gov/mdhhs/inside-mdhhs/newsroom/2026/01/06/vaccines-state
MDHHS issues statement about federal changes to childhood vaccine schedule
LANSING, Mich. – Following updates to the U.S. childhood immunization schedule made today by Deputy Secretary of Health and Human Services Jim O’Neill, in his role as acting director of the Centers for Disease Control and Prevention, the Michigan Department of Health and Human Services issued the following statement:
“For decades, vaccines have played a critical role in the prevention and control of infectious diseases and significant reductions in childhood illnesses and fatalities.
On Thursday, Dec. 18, Michigan’s Chief Medical Executive Dr. Natasha Bagdasarian issued a Standing Recommendation advising health care providers and families to follow the child and adolescent immunization schedule produced by the American Academy of Pediatrics (AAP) or the American Academy of Family Physicians (AAFP). We continue to stand by that recommendation.”
The underlying scientific evidence remains unchanged and continues to support the full AAP and AAFP vaccination schedules for children. Families should still be able to access the full range of childhood immunizations as recommended by the AAP and AAFP to protect their children from serious diseases.
All vaccines, including those moved to shared clinical decision-making, remain covered with no out-of-pocket cost by Affordable Care Act-regulated private insurance plans and federal coverage programs such as Medicaid and the Vaccines for Children program, as HHS affirmed in its announcement.
Bagdasarian noted that the changes announced today may create confusion for families and clinicians regarding school vaccine requirements, clinical workflows and the supply and use of combination vaccines.
“MDHHS will continue to provide clear guidance, backed by science to help protect Michigan families,” Bagdasarian said.
# # #
Notice the federal/state divide?
The state is allying with private organizations. Parents are more free to make healthcare choices.
I love the direction this is going for healthcare freedom.
Get MHF Insights
News and tips for your healthcare freedom.
We never spam you. One-step unsubscribe.