- When your AI agent goes off script: What Moffitt Cancer Center caught early
- Utah physician, 2 nurses charged with $29M fraud scheme
- 3 Ascension CFO moves in 2 days
- The volume paradox costing ASCs millions
- Pediatric radiology leaders launch AI brain tumor dataset
- Inside SCA Health’s clinical nursing ladder — and how it helped cut turnover by 7%
- What ASCs are getting wrong about OR delays — and how to fix it
- 1 behavioral health leader’s playbook for staff retention and safety
- Centene executive to join Froedtert ThedaCare as chief nurse
- Behavioral health ED visits projected to rise by 1 million
- UPMC provides free RN-BSN degree to its nurses
- Physicians aren’t buying payers’ prior auth pledge: 5 survey notes
- 5 dental AI updates in 1 month
- 3 lawsuit settlements in dentistry
- Financial pressures shutter Iowa dental clinics: 4 notes
- 4 cities where systems are winning new grads from tech, finance
- Medline launches predictive supply chain platform and 5 more updates to know
- Independent autism research committee adds 7 members
- FDA Commissioner Marty Makary Resigns After Trump Pressure
- 10 notes on the widening DSO performance gap
- Health systems are betting on retail partnerships
- CDC-linked autism researcher arraigned on fraud charges
- Nearly 1 in 3 boys under 14 discussed suicide in crisis conversations: Report
- How anesthesia became a financial liability for ASCs
- 4 pressures converging on pharmacy
- Ascension Texas hospital taps new assistant CFO
- 82% of physicians are now employed: 6 notes
- Atlanta-area medical office portfolio sold in leaseback deal
- New warning sign for sudden cardiac death found: Study
- NAPA partners with New York hospital
- Remarks at the MFA Legal & Compliance 2026 Conference
- GLP-1 Drugs May Improve Breast Cancer Outcomes
- CMS pauses hospice, home health Medicare enrollments in fraud crackdown
- Merck KGaA looks to M&A to bolster its 'rather slim' pipeline
- Takeda, slimming down for 'new era,' plots 4,500 layoffs in latest restructuring drive
- BeOne Medicines’ Brukinsa TV ad 'Clarity' hit by FDA over 'misleading suggestions'
- Health systems are exploring AI-powered opportunistic screening. New CMS reimbursement could unlock a business case for it
- Most mental health practitioners satisfied with work, financially stable, SimplePractice finds
- Ted Turner's Brain Disease More Common Than Previously Thought, Review Finds
- Novo, Lilly tout respective early response and weight loss maintenance data as GLP-1 rivalry intensifies
- Ivermectin Prescriptions Doubled After Mel Gibson Cancer Cure Claim
- Haleon tackles sports injuries with latest soccer play
- Eating Out Linked To Obesity Risk Worldwide
- Perimenopausal Women Face Greater Heart Risk, Study Finds
- Low Wages, Empty Plates, Heavy Toll: Rethinking Suicide Prevention
- RFK Jr. Swaps Vaccine Talk for Healthy Foods and Reading to Tots in Push To Woo Voters
- Trump and Kennedy Seek To Relax Safeguards for AI Healthcare Tools
- Valneva to lay off up to 15% of workforce in face of ‘adverse trend’ in travel vaccines
- California to award $111M for behavioral health supportive housing
- 6 new psychiatric residency programs to know
- USOSM adds New York practice
- NAMI partners on health crisis preparation hub
- Oklahoma enacts law expanding access to dental care
- Where dentists are leaving value behind in practice sales
- Why dental practices are closing in 2026
- Texas dental school to launch master’s program with orthodontic specialty
- Qualitas Dental Partners makes investments into 7 practices
- Mayo Clinic CEO Gianrico Farrugia stepping down at year's end
- 988 calls are rising — what’s behind the surge?
- Providence hospital to lay off 40 workers amid behavioral health staffing overhaul
- BioMarin consolidates staff at Amicus HQ after closing $4.8B deal for rare disease peer
- US Monitors For Hantavirus As WHO Expects More Cases But 'Not Another COVID'
- University of California, union near May 14 strike deadline with no deal in hand
- 1 in 5 marketplace enrollees dropped their coverage in 2026: media report
- Hims & Hers posts $92M loss in Q1 as it shifts to branded GLP-1 medications
- Listen to the Latest ‘KFF Health News Minute’
- FDA Commissioner Marty Makary to resign, capping turbulent tenure
- FDA Commissioner Marty Makary to resign, capping turbulent tenure
- Providence puts years of losses in rearview with its third consecutive quarter of operating gains
- Millions of Women Suffer in Silence From Treatable Pelvic Organ Prolapse
- Eli Lilly pauses Indian obesity awareness campaign after regulatory notices: report
- Optum Rx unveils new transparent PBM model
- Fitness wearable Whoop adds on-demand clinician access, EHR syncing
- Alkermes’ Lumryz hits phase 3 mark in another sleep disorder, fueling momentum from $2.4B Avadel acquisition
- ACA exchanges take spotlight in Q1
- Pfizer, Arvinas win $85M upfront in Rigel licensing pact for new breast cancer med Veppanu
- Bayer's Eylea declines by 24%, bearing the brunt of biosmilar competition
- As public vaccine criticism quiets, RFK Jr. keeps safety inquiries running in background: NYT
- As public vaccine criticism quiets, RFK Jr. keeps safety inquiries running in background: NYT
- What's Fueling The High U.S. Death Rate? It Might Not Be What You Think
- Telemedicine Not Breaking The Bank, Also Not Expanding Patient Access
- After-School Sports An Overall Boon To Children And Teens, Study Shows
- Trump Promised Cheaper Drugs. Some Prices Dropped. Many Others Shot Up
- Why Are Older Adults Taking Edibles? Survey Reveals Some Surprises
- Low Wages, Empty Plates, Heavy Toll: Rethinking Suicide Prevention
- EU advances scheme to bolster manufacturing autonomy, avert drug shortages
- Bicara Therapeutics hires Replimune, Sanofi alum as chief commercial officer
- The broken pipeline of mental healthcare for LGBTQ teenagers
- FDA Launches One-Day Inspectional Assessments to Strengthen and Expand Oversight
- FDA Launches One-Day Inspectional Assessments to Strengthen and Expand Oversight
- Is your hospital ready for a prolonged IT outage? Joint Commission, AHA's new resiliency program will let you know
- FDA Expands AI Capabilities: Launches ELSA and Completes HALO Data Platform Consolidation
- FDA Expands AI Capabilities: Launches ELSA and Completes HALO Data Platform Consolidation
- Roche acquires PathAI to transform AI-driven diagnostics
- Roche acquires PathAI to transform AI-driven diagnostics
- Trump Planning to Fire FDA Commissioner Marty Makary
- Trump Planning to Fire FDA Commissioner Marty Makary
- Included Health launches AI-powered solution to connect members to providers
- FDA Green Lights Bizengri Drug To Treat Rare, Aggressive Bile Duct Cancer
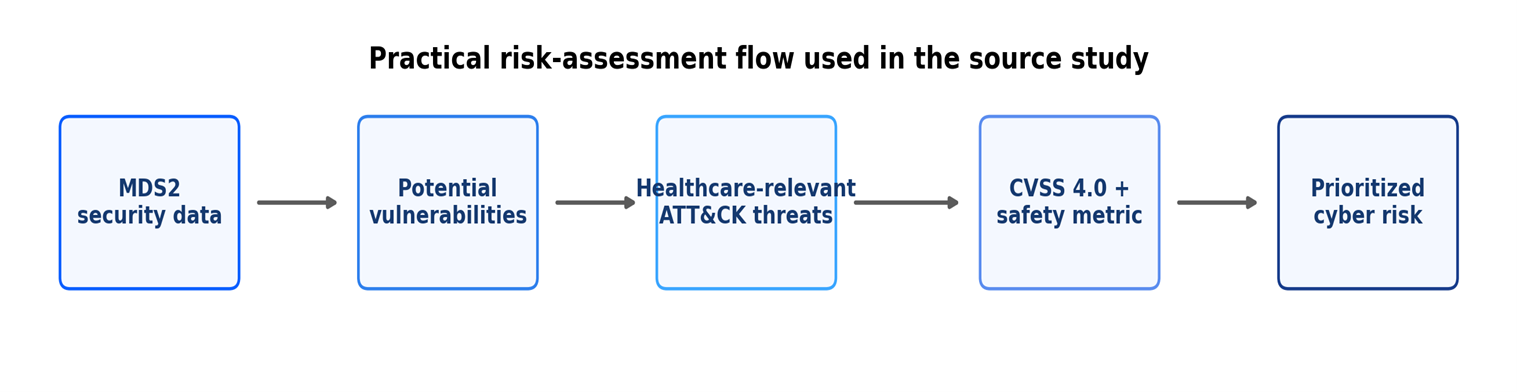
- The Hidden Design Flaw in Medical Device Service Technology
- The Hidden Design Flaw in Medical Device Service Technology
- An Endovascular Approach to Neurological Diseases Can Shift the Treatment Paradigm
- An Endovascular Approach to Neurological Diseases Can Shift the Treatment Paradigm
- 8,500 Steps A Day Could Be Sweet Spot For Preventing Weight Regain
- Why Gen AI is a Win for MedTech: And, How to Unlock its Potential with the Right Policies
- Why Gen AI is a Win for MedTech: And, How to Unlock its Potential with the Right Policies
- Survey: Employers seeking greater transparency from pharmacy benefits
- Kaiser Permanente's investments pick up the slack as Q1 operating margin slims to 2.1%
- AMA unveils policy framework to combat AI deepfake physician impersonation
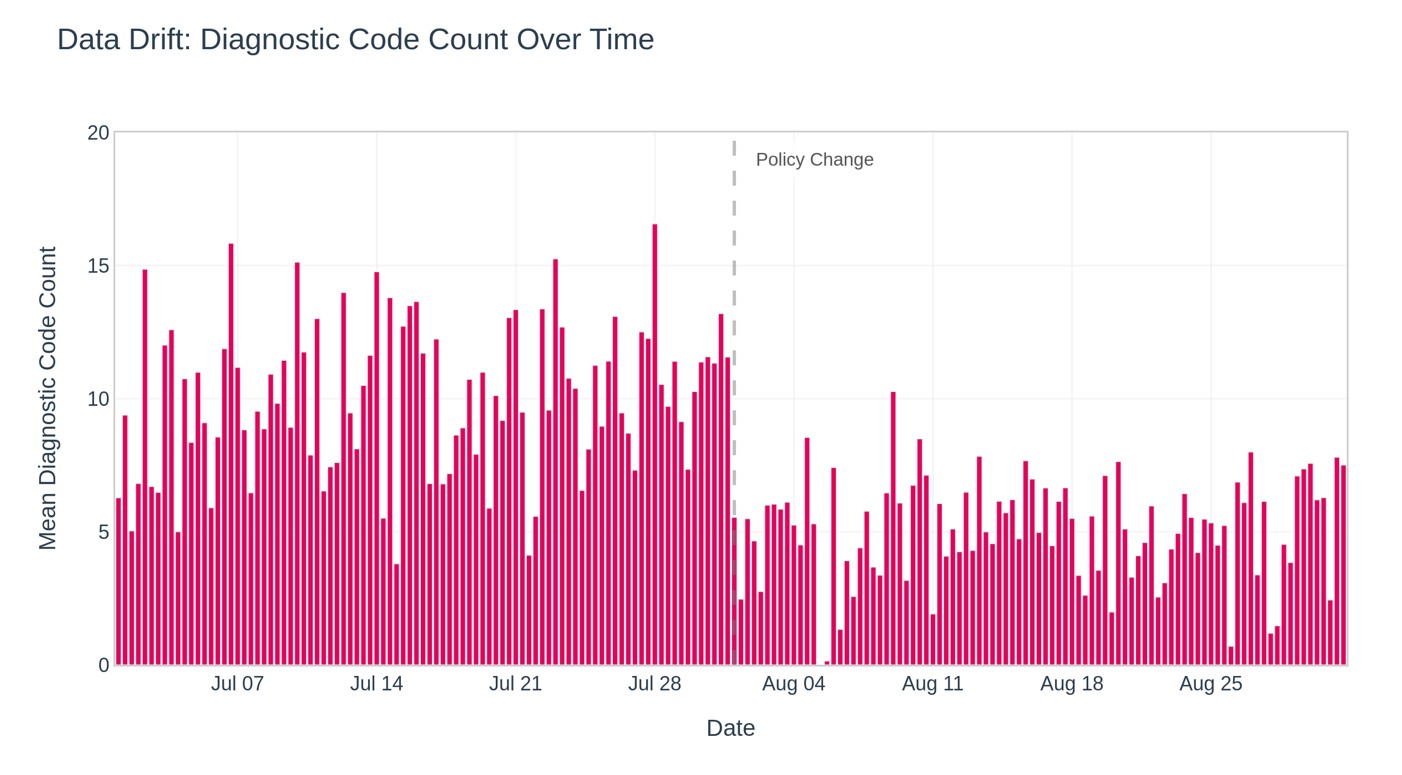
- The Medical Device Cybersecurity Gap Hiding in Plain Sight
- The Medical Device Cybersecurity Gap Hiding in Plain Sight
- CSL slashes revenue projection and takes $5B impairment as interim CEO flags R&D misses, market erosion
- Healthcare bankruptcies up 33% in Q1 2026: report
- Why Doctors Are Quitting At An Earlier Age
- Sharper Brains May Face Higher Depression Relapse Risk, Study Finds
- Older Adults Have Fewer Regrets, Study Says
- Partner's bispecific Bizengri nabs FDA national priority nod in rare bile duct cancer
- Daiichi Sankyo targets global top 5 oncology rank by 2035, $1.3B efficiency drive in new 5-year plan
- That Discount At The Pharmacy Counter May Pack Hidden Costs
- Nighttime Heat Waves Increase Asthma Risk
- Watch: 8 Health Insurance Terms You Should Know
- As Ranks of Uninsured Grow, Minnesota’s Hospitals Are Among Least Charitable in Nation
- OVID Health hires Edelman alum Davide Scalenghe to boost its international footprint
- Maintaining trust in medical AI: Monitoring and managing model lifecycle
- Maintaining trust in medical AI: Monitoring and managing model lifecycle
- Eli Lilly shoots for health in new Caitlin Clark ad campaign
- Omada Health posts 42% revenue jump in Q1, joins Eli Lilly employer weight loss program
- Journalists Shed Light on Deadly Hantavirus Outbreak and a Crisis in the Nation’s ERs
- The Make America Healthy Again Movement Comes for Hospital Food
- Remarks at the Conference on Financial Market Regulation
- Dad Jokes: Remarks at the 13th Annual Conference on Financial Markets Regulation
- RFK Jr. Launches Plan To Curb Antidepressant 'Overprescription'
- Skil-Care launches specialized healthcare product innovation program
- Sanford Health unveils deal to integrate Minnesota-area North Memorial Health, invest $600M
- UPDATED—Trump plans to fire FDA chief Marty Makary: report
- Remarks at the Special Competitive Studies Project AI+ Expo
- Plant-Based Foods May Help Lower Risk of High Blood Pressure
- Integrated CDO capabilities reduce early development complexity
- Targeted Protein Degradation and Novel Modalities: Getting on the Frontline
- Workplace safety is a top priority for 93% of healthcare leaders: Axon survey
- Super Shoes Might Increase Risk Of Running Injuries, Study Says
- TV, Movies Offer Flawed Depictions Of Autism, Add To Delayed Diagnosis, Study Says
- Opioid OD Survivors Have Triple Rate Of Repeat Overdoses Than Previously Estimated
- A New Medicare Option For Weight Loss Drugs: What Older Americans Should Know
- Exposure Therapy Can Successfully Ease Peanut Allergies
- Listen: A Federal Agency Is After Workers’ Health Data, and Critics Are Alarmed
- In California Governor Race, Single-Payer Is a Litmus Test. There’s Still No Way To Pay for It.
- Cruise Ship Hantavirus Outbreak Kills 3 as WHO Says Risk Is Low
- Remarks at the 13th Annual Conference on Financial Market Regulation
- New Study Suggests The Brain Can Continue Learning While In An Unconscious State

Stat News guest opinion author sounds off against recent policy changes.
Coming from within Big Pharma, he has certainly benefited from federal funds.
Wonder if he realizes that the same Constitution which would grant his Patent concerns, would (read in its original sense) do more than approve his FDA and Medicare price-fixing complaints. It would eliminate both programs, departments, and industry funding from federal government!
https://www.statnews.com/2023/07/06/biden-cancer-moonshot-ira-intellectual-property/
July 6, 2023When then-Vice President Biden launched the Cancer Moonshot in 2016, there was reason to be optimistic. Decades of rigorous science had transformed many cancers from a death sentence to a manageable chronic disease. New treatment advances — including gene and cell therapies and immunotherapies — were showing stunning results. With support from the biopharmaceutical industry and the help of smart government policies, ending cancer was, and is, an achievable goal.
Unfortunately, while the president promised a war on cancer, he declared a war on the cure instead. As the head of PhRMA, an association representing America’s leading biopharmaceutical research companies, I work with my team to closely follow the policy debate and the impact on innovation. While they may have been well-intended, the Biden administration has adopted a series of policies that attack critical steps needed to bring new treatments out of the lab and to the patients who need them.
First, the administration eroded the foundation of scientific innovation by subverting intellectual property protections. In 2022, the White House caved to foreign competitors and global activists by agreeing at the World Trade Organization ministerial conference to waive IP protections that enabled the development of Covid-19 vaccines.
That caused the first tear in the IP fabric, signaling the United States would no longer vigorously defend the IP rights of researchers. Countries including China and India are now angling to waive IP protections for Covid-19 treatments, including those that could potentially treat cancer and other diseases. The administration has been unwilling to stop this global IP heist.
For life sciences companies, IP is the foundation. The certainty of knowing it will have exclusive rights for a limited time before copies hit the market is what enables companies to make long-term, risky investments into new medicines.Next, the administration signed legislation giving them the authority to unilaterally determine the value of innovative medicines. Under the guise of “negotiation,” the Inflation Reduction Act (IRA) allows the federal government to set the price of medicines in Medicare.
Every time it has been tried around the world, government price setting reduces patient access to new cures and treatments. According to a recent report we released, patients in the U.S. have access to 85% of new medicines compared with 43% in France and 21% in Canada where governments set the price. Government price setting also leads to less innovation, and in the case of the IRA, cancer research will be especially hard-hit.
For example, the IRA’s price setting provisions disproportionately impact small-molecule medicines — medicines that typically come in pill or tablet form. These small but mighty treatments have a unique ability to target specific processes inside cells that allow tumors to grow and spread. The majority of cancer medicines approved for use today are small molecules.
Medicines typically face generic competition after they’ve been on the market 14 years. But the IRA cuts that time to nine years after FDA approval for small-molecule treatments. The consequences of this “pill penalty” are predictable. In a survey of our member companies, 63% who responded said they plan to shift focus away from small-molecule medicines.
The law’s price setting scheme also threatens research on existing treatments. Research companies often conduct clinical trials to learn whether an already-approved treatment can help more patients with a similar disease or those with different diseases entirely. It can take years to get results that show an approved treatment could benefit other patients.
Consider Xalkori. This targeted oral cancer therapy was first approved in 2011 for the treatment of an advanced non-small cell lung cancer caused by a specific gene abnormality. It would later receive six additional approvals across several patient populations, including one more than a decade after its initial approval.
This is how innovation works. A new analysis that the Partnership for Health Analytic Research prepared for PhRMA found that 61% of small-molecule cancer treatments approved between 2006 and 2012 received additional approvals for indications to treat other forms of cancers, with 41% occurring seven years or more after initial approval.
This post-approval research has been critical in the fight against cancer, as well as pediatric and rare diseases. But if it takes seven years to bring these treatments across the finish line and the government sets the price two years later, then it disincentivizes the clinical research needed to develop these new therapies.
As it writes rules to implement the IRA, the administration is making a bad law worse. Once a treatment is selected for government price setting, the administration intends to sweep in every new form that followed a medicine’s initial approval. This will subject to price controls new treatments that have been available for just a few years or even less.
Finally, the Biden administration is threatening the availability of medicines approved by the FDA through a program that has provided timely access to hundreds of treatments. Accelerated approval has sped the development of new medicines for patients where unmet need is greatest and no alternative or viable treatments exist, while adhering to the same standards for safety and effectiveness as traditional approval. It has been a resounding success since it was first developed during the HIV/AIDS crisis.
Fast forward to today, and the administration is supporting policies that would limit Medicare payment for medicines granted accelerated approval. Once again, those with cancer will be disproportionately harmed. Nearly 70% of medicines with accelerated approval have been in oncology. Companies will be forced to abandon the program if patients cannot access these treatments, and potentially lifesaving medicines will take longer to reach patients than they otherwise would.
The cumulative effect of these misguided decisions will be felt for years to come. They will undermine America’s global leadership and competitiveness. And they will harm our ability to develop new treatments for a whole host of diseases.
Advances in cancer research are not accidents, nor are they inevitable. They emerge from an ecosystem that encourages risk, discipline and drive — one we must preserve and protect. President Biden should end the assault on that ecosystem, and in doing so, help win the fight against cancer.
Steve Ubl is the president and CEO of PhRMA.
Get MHF Insights
News and tips for your healthcare freedom.
We never spam you. One-step unsubscribe.