- 8 health systems, more than $1.4B in capital projects in May
- RFK Jr. visits Summa Health
- Kaiser Permanente plans 11K-square-foot MOB in Nevada
- Dentistry’s AI gold rush
- Community Health Network taps new CFO from Aspirus Health
- California creates $25M fund for at-risk hospitals
- Only 1 in 5 physicians is independent: 10 new numbers behind the collapse
- California physician group acquired by PRISM
- Behavioral health ED visits projected to rise by 1 million
- When your AI agent goes off script: What Moffitt Cancer Center caught early
- Utah physician, 2 nurses charged in $29M fraud scheme
- 3 Ascension CFO moves in 2 days
- The volume paradox costing ASCs millions
- Pediatric radiology leaders launch AI brain tumor dataset
- Inside SCA Health’s clinical nursing ladder — and how it helped cut turnover by 7%
- What ASCs are getting wrong about OR delays — and how to fix it
- 1 behavioral health leader’s playbook for staff retention and safety
- Centene executive to join Froedtert ThedaCare as chief nurse
- Behavioral health ED visits projected to rise by 1 million
- UPMC provides free RN-BSN degree to its nurses
- Physicians aren’t buying payers’ prior auth pledge: 5 survey notes
- 5 dental AI updates in 1 month
- 3 lawsuit settlements in dentistry
- Financial pressures shutter Iowa dental clinics: 4 notes
- Medline launches predictive supply chain platform and 5 more updates to know
- Independent autism research committee adds 7 members
- FDA Commissioner Marty Makary Resigns After Trump Pressure
- 10 notes on the widening DSO performance gap
- CDC-linked autism researcher arraigned on fraud charges
- Nearly 1 in 3 boys under 14 discussed suicide in crisis conversations: Report
- How anesthesia became a financial liability for ASCs
- As Trump arrives in China, Big Pharma CEOs are notably absent
- Remarks at the MFA Legal & Compliance 2026 Conference
- GLP-1 Drugs May Improve Breast Cancer Outcomes
- CMS pauses hospice, home health Medicare enrollments in fraud crackdown
- NYU Langone Health says it received grand jury subpoena over gender-affirming care
- Merck KGaA looks to M&A to bolster its 'rather slim' pipeline
- Takeda, slimming down for 'new era,' plots 4,500 layoffs in latest restructuring drive
- BeOne Medicines’ Brukinsa TV ad 'Clarity' hit by FDA over 'misleading suggestions'
- Health systems are exploring AI-powered cardiac risk screening. New CMS reimbursement could unlock a business case for it
- Most mental health practitioners satisfied with work, financially stable, SimplePractice finds
- Ted Turner's Brain Disease More Common Than Previously Thought, Review Finds
- Novo, Lilly tout respective early response and weight loss maintenance data as GLP-1 rivalry intensifies
- Ivermectin Prescriptions Doubled After Mel Gibson Cancer Cure Claim
- Haleon tackles sports injuries with latest soccer play
- Eating Out Linked To Obesity Risk Worldwide
- Perimenopausal Women Face Greater Heart Risk, Study Finds
- Low Wages, Empty Plates, Heavy Toll: Rethinking Suicide Prevention
- RFK Jr. Swaps Vaccine Talk for Healthy Foods and Reading to Tots in Push To Woo Voters
- Trump and Kennedy Seek To Relax Safeguards for AI Healthcare Tools
- Valneva to lay off up to 15% of workforce in face of ‘adverse trend’ in travel vaccines
- California to award $111M for behavioral health supportive housing
- 6 new psychiatric residency programs to know
- USOSM adds New York practice
- NAMI partners on health crisis preparation hub
- Oklahoma enacts law expanding access to dental care
- Where dentists are leaving value behind in practice sales
- Why dental practices are closing in 2026
- Texas dental school to launch master’s program with orthodontic specialty
- Mayo Clinic CEO Gianrico Farrugia stepping down at year's end
- 988 calls are rising — what’s behind the surge?
- Providence hospital to lay off 40 workers amid behavioral health staffing overhaul
- BioMarin consolidates staff at Amicus HQ after closing $4.8B deal for rare disease peer
- US Monitors For Hantavirus As WHO Expects More Cases But 'Not Another COVID'
- University of California, union near May 14 strike deadline with no deal in hand
- 1 in 5 marketplace enrollees dropped their coverage in 2026: media report
- Hims & Hers posts $92M loss in Q1 as it shifts to branded GLP-1 medications
- Listen to the Latest ‘KFF Health News Minute’
- FDA Commissioner Marty Makary to resign, capping turbulent tenure
- FDA Commissioner Marty Makary to resign, capping turbulent tenure
- Providence puts years of losses in rearview with its third consecutive quarter of operating gains
- Millions of Women Suffer in Silence From Treatable Pelvic Organ Prolapse
- Eli Lilly pauses Indian obesity awareness campaign after regulatory notices: report
- Optum Rx unveils new transparent PBM model
- Fitness wearable Whoop adds on-demand clinician access, EHR syncing
- Alkermes’ Lumryz hits phase 3 mark in another sleep disorder, fueling momentum from $2.4B Avadel acquisition
- ACA exchanges take spotlight in Q1
- Bayer's Eylea declines by 24%, bearing the brunt of biosmilar competition
- Pfizer, Arvinas win $85M upfront in Rigel licensing pact for new breast cancer med Veppanu
- As public vaccine criticism quiets, RFK Jr. keeps safety inquiries running in background: NYT
- As public vaccine criticism quiets, RFK Jr. keeps safety inquiries running in background: NYT
- What's Fueling The High U.S. Death Rate? It Might Not Be What You Think
- Telemedicine Not Breaking The Bank, Also Not Expanding Patient Access
- After-School Sports An Overall Boon To Children And Teens, Study Shows
- Trump Promised Cheaper Drugs. Some Prices Dropped. Many Others Shot Up
- Why Are Older Adults Taking Edibles? Survey Reveals Some Surprises
- Low Wages, Empty Plates, Heavy Toll: Rethinking Suicide Prevention
- EU advances scheme to bolster manufacturing autonomy, avert drug shortages
- Bicara Therapeutics hires Replimune, Sanofi alum as chief commercial officer
- The broken pipeline of mental healthcare for LGBTQ teenagers
- FDA Launches One-Day Inspectional Assessments to Strengthen and Expand Oversight
- FDA Launches One-Day Inspectional Assessments to Strengthen and Expand Oversight
- Is your hospital ready for a prolonged IT outage? Joint Commission, AHA's new resiliency program will let you know
- FDA Expands AI Capabilities: Launches ELSA and Completes HALO Data Platform Consolidation
- FDA Expands AI Capabilities: Launches ELSA and Completes HALO Data Platform Consolidation
- Roche acquires PathAI to transform AI-driven diagnostics
- Roche acquires PathAI to transform AI-driven diagnostics
- Trump Planning to Fire FDA Commissioner Marty Makary
- Trump Planning to Fire FDA Commissioner Marty Makary
- Included Health launches AI-powered solution to connect members to providers
- FDA Green Lights Bizengri Drug To Treat Rare, Aggressive Bile Duct Cancer
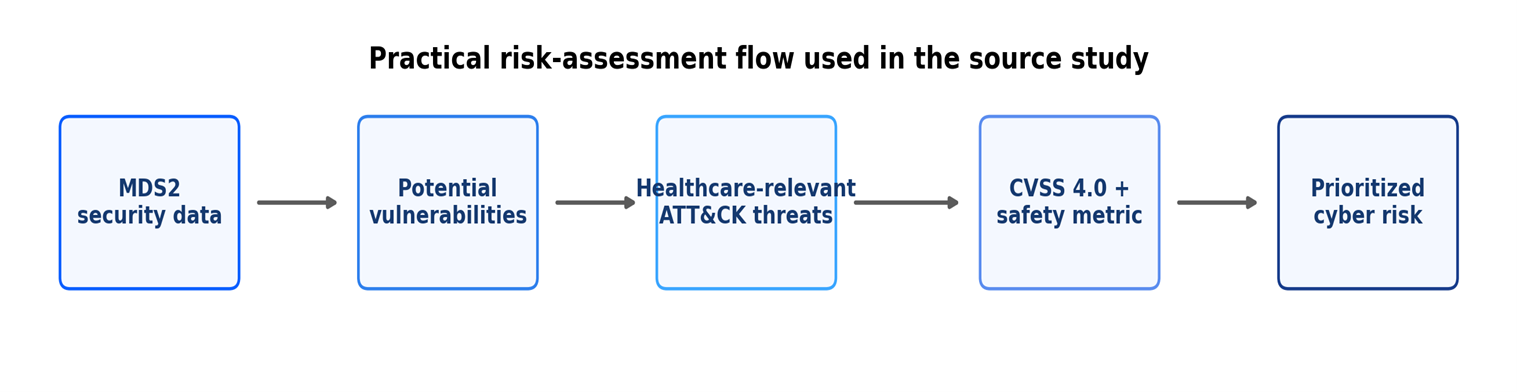
- The Hidden Design Flaw in Medical Device Service Technology
- The Hidden Design Flaw in Medical Device Service Technology
- An Endovascular Approach to Neurological Diseases Can Shift the Treatment Paradigm
- An Endovascular Approach to Neurological Diseases Can Shift the Treatment Paradigm
- 8,500 Steps A Day Could Be Sweet Spot For Preventing Weight Regain
- Why Gen AI is a Win for MedTech: And, How to Unlock its Potential with the Right Policies
- Why Gen AI is a Win for MedTech: And, How to Unlock its Potential with the Right Policies
- Survey: Employers seeking greater transparency from pharmacy benefits
- Kaiser Permanente's investments pick up the slack as Q1 operating margin slims to 2.1%
- AMA unveils policy framework to combat AI deepfake physician impersonation
- The Medical Device Cybersecurity Gap Hiding in Plain Sight
- The Medical Device Cybersecurity Gap Hiding in Plain Sight
- CSL slashes revenue projection and takes $5B impairment as interim CEO flags R&D misses, market erosion
- Healthcare bankruptcies up 33% in Q1 2026: report
- Why Doctors Are Quitting At An Earlier Age
- Sharper Brains May Face Higher Depression Relapse Risk, Study Finds
- Older Adults Have Fewer Regrets, Study Says
- Partner's bispecific Bizengri nabs FDA national priority nod in rare bile duct cancer
- Daiichi Sankyo targets global top 5 oncology rank by 2035, $1.3B efficiency drive in new 5-year plan
- That Discount At The Pharmacy Counter May Pack Hidden Costs
- Nighttime Heat Waves Increase Asthma Risk
- As Ranks of Uninsured Grow, Minnesota’s Hospitals Are Among Least Charitable in Nation
- Watch: 8 Health Insurance Terms You Should Know
- OVID Health hires Edelman alum Davide Scalenghe to boost its international footprint
- Maintaining trust in medical AI: Monitoring and managing model lifecycle
- Maintaining trust in medical AI: Monitoring and managing model lifecycle
- Eli Lilly shoots for health in new Caitlin Clark ad campaign
- Omada Health posts 42% revenue jump in Q1, joins Eli Lilly employer weight loss program
- Journalists Shed Light on Deadly Hantavirus Outbreak and a Crisis in the Nation’s ERs
- The Make America Healthy Again Movement Comes for Hospital Food
- Remarks at the Conference on Financial Market Regulation
- Dad Jokes: Remarks at the 13th Annual Conference on Financial Markets Regulation
- RFK Jr. Launches Plan To Curb Antidepressant 'Overprescription'
- Skil-Care launches specialized healthcare product innovation program
- Remarks at the Special Competitive Studies Project AI+ Expo
- Plant-Based Foods May Help Lower Risk of High Blood Pressure
- Integrated CDO capabilities reduce early development complexity
- Targeted Protein Degradation and Novel Modalities: Getting on the Frontline
- Workplace safety is a top priority for 93% of healthcare leaders: Axon survey
- Super Shoes Might Increase Risk Of Running Injuries, Study Says
- TV, Movies Offer Flawed Depictions Of Autism, Add To Delayed Diagnosis, Study Says
- Opioid OD Survivors Have Triple Rate Of Repeat Overdoses Than Previously Estimated
- A New Medicare Option For Weight Loss Drugs: What Older Americans Should Know
- Exposure Therapy Can Successfully Ease Peanut Allergies
- Listen: A Federal Agency Is After Workers’ Health Data, and Critics Are Alarmed
- In California Governor Race, Single-Payer Is a Litmus Test. There’s Still No Way To Pay for It.
- Cruise Ship Hantavirus Outbreak Kills 3 as WHO Says Risk Is Low
- Remarks at the 13th Annual Conference on Financial Market Regulation
- New Study Suggests The Brain Can Continue Learning While In An Unconscious State

President Trump is determined to drive the pharmaceutical industry out of China:
https://thehill.com/homenews/administration/5239938-trump-major-tariffs-pharmaceuticals/
Trump: ‘Major’ pharmaceutical tariffs coming soon
By Ashleigh Fields - April 9, 2025President Trump said Tuesday his administration soon would announce “major” tariffs on pharmaceuticals, adding to the tariffs he has already imposed on most goods entering the United States from abroad.
“We’re going to be announcing very shortly a major tariff on pharmaceuticals,” Trump said during a speech at the National Republican Congressional Committee.
“And when they hear that, they will leave China. They will leave other places because they have to sell — most of their product is sold here, and they’re going to be opening up their plants all over the place in our country,” he added.
Since 2020, U.S. imports of Chinese pharmaceuticals — defined by the U.S. tariff code to include packaged medicaments, vaccines, blood, organic cultures, bandages, and organs — have grown by 390 percent, going from $2.1 billion in 2020 to $10.3 billion in 2022 according to the Atlantic Council.
The country has streamlined biotech production in the capital of Beijing while cutting costs for production after launching a five-year plan to increase the development of consumer drugs.
Trump’s proposed trade policies aim to hamper China’s pharmaceutical growth by encouraging plants to set up shop in the U.S. due to the onset of high levies.
“What other presidents allow China to get away with is absolutely criminal, but I’m not like the other presidents, and it’s not going to happen under President Trump. It’s just not going to happen,” he told his GOP colleagues on Tuesday.
The leader said China’s retaliation to the tariffs he announced last week has further fueled his plan to slam their supply chain growth.
China announced a 34 percent tariff on U.S. goods last week in response to Trump tariffs imposed on Beijing. This week, each country has added a 50 percent tariff on the other’s goods.
“After all of the abuses they’ve perpetrated, China’s attempting to impose additional unjustified tariffs. Just so you understand, they all got rich because of tariffs, you know. Now when we do it, oh, it’s so terrible. What Trump is doing, it’s so terrible. They got rich,” the president said.
“And not only China, many countries.”
By citing complex US pharmaceutical sourcing problems today, Morning Brew tacitly acknowledges the wisdom of pharma tariffs.
https://www.healthcare-brew.com/stories/2025/04/11/pharma-tariffs-impact-healthcare
Pharma tariffs are coming ‘very shortly.’ Here’s how they could impact healthcare
Companies are likely to see cost increases despite an initial exemption.
By Cassie McGrath | April 11, 2025The pharmaceutical industry seemingly came out on top when President Donald Trump announced import tariffs on April 2. And while those are currently on a 90-day pause for every country except China, giving a break to industries like auto and steel, it seems the shoe might be on the other foot for pharma now.
Under the original plan, those industries faced at least 10% tariffs for goods imported to the US, kicking off a stock market drop. But pharma had been excluded, and major players’ stocks responded positively.
Companies like Johnson & Johnson and Eli Lilly saw small bumps (1.4% and 1.6%, respectively from April 1 to 2) as the rest of the market suffered. Pharma was exempted because the industry’s supply chain is spread out and complicated, and also because the medication it makes can be lifesaving.
“I was encouraged that they were exempted from tariffs, just given the essential nature of pharmaceuticals and also the complexity of the pharmaceutical supply chain,” Evan Seigerman, biopharma analyst at investment bank BMO Capital Markets, told Healthcare Brew. “You can’t just easily move manufacturing from one jurisdiction to another.”
But when announcing the pause on April 9, Trump also said the grace period for pharma could end “very shortly,” which caused the pharma market to dip. By around noon on April 10, J&J’s stock was down 13% and Lilly’s dropped 5%.
Seigerman and his colleagues wrote in an April 9 report they’re “strongly opposed” to tariffs for pharmaceuticals because the strategy is unlikely to bring manufacturing back to the US. “Shock [to] the pharma supply chain threatens our own public health and access to medicines—from generic antibiotics to complex biologic and peptides,” they wrote. “The ongoing gutting of [the] FDA is bad enough, only now to be compounded by what we believe to be real talk of pharma tariffs.”
Bringing it home
When Trump announced the tariff pause, he said “[pharmaceutical companies] will leave other places because they have to sell—most of their product is sold here and they’re going to be opening up their plants all over the place.”
According to the FDA, the number of manufacturing facilities in China making active pharmaceutical ingredients doubled from 2010 to 2019.
Roche, a pharmaceutical company with more than 25,000 US employees, has an “entire pharma and diagnostics value chain” in the country already, with four R&D, production, and distribution sites in the pharmaceutical division as well as seven R&D and manufacturing sites for diagnostics, according to Nina Maehlitz, the company’s global media spokesperson.
Still, Maehlitz said it’s “considering additional US investments to continue to meet patients’ needs.”
Eric Axel, executive director at American Medical Manufacturers Association, which represents companies that make medical supplies in the US like masks and gloves, agreed that more products should be made domestically.
A 2021 report from the FDA’s Center for Drug Evaluation and Research Side Catalog found that 46% of manufacturing facilities that make medications and 26% that make active pharmaceutical ingredients for drugs taken in the US are made domestically.
“If we rely too much on foreign suppliers, we leave ourselves at the mercy of these foreign countries, which a lot of times do not have our best interests at hand,” he said, adding that there are many domestic manufacturers who want business from companies “complaining” that their supply chain has been disrupted.
Cost concerns
Plus, Seigerman said, costs are likely to rise if other tariffs go back into effect, as the prices of non-drug goods used in research and development—like microscopes, glass bottles, electronic equipment, and raw materials—may go up.
Eli Lilly declined to comment, but CEO David Ricks told the BBC if the company does face rising costs, it may have to make cuts, likely in staff and in R&D research and development spending.
But other experts said there are benefits to making drugs in other countries, namely the environmental impacts. Among these issues is the rise in animal poisonings, contaminated algae, and drug-resistant bacteria, according to the European Environmental Bureau.
“A lot of the processes to make the API, or active pharmaceutical ingredients, are not really environmentally friendly processes,” Seigerman said.
Uncertain future
On Feb. 18, for example, Trump said pharmaceuticals could see tariffs upward of 25% or more that would increase over the year, but he wanted to “give them time to come” back to the US.
Companies like Eli Lilly and Johnson & Johnson have already committed billions of dollars ($50 billion and $55 billion, respectively) to reshoring. J&J plans to open three facilities, including one that is already under construction in North Carolina. Lilly is planning four manufacturing sites in North Carolina and Wisconsin as well as expansions in Indiana.
Pankit Bhalodia, a partner in life sciences at consulting firm West Monroe, said it’s hard for many companies to make decisions amid the uncertainty.
On April 3, West Monroe released the results of a survey across all industries that showed tariffs, geopolitical tensions, and cost of materials were among supply chain executives’ top concerns in Q1.
“Whether it’s large or small pharmaceutical companies, the supply chain is very complex, very distributed, very tangled with a lot of countries,” Bhalodia said. “It’s not truly exempt as it was perceived.”
Two European-based reporters at Politico EU say American bureaucracy and foreign retention efforts will frustrate President Trump's tariff drive to reshore pharmaceutical manufacturing:
https://www.politico.eu/article/us-donald-trump-pharma-tariffs-drug-production/
Trump says pharma tariffs will entice back drug production. They won’t.
Pharmaceuticals were the EU’s largest export to the U.S. last year, worth a reported $127B.
By Mari Eccles and Hanne Cokelaere - April 14, 2025In 2024, pharmaceuticals were the EU’s largest export to the U.S., worth a reported $127 billion. | Ritesh Shukla/Getty Images
BRUSSELS — After hitting just about every other industry he can think of (albeit with a brief pause), Donald Trump’s long-promised tariffs on drugs are around the corner.
“We’re going to be announcing very shortly a major tariff on pharmaceuticals,” he said at a dinner of the National Republican Congressional Committee last week.
“And when they hear that, they will leave China. They will leave other places because they have to sell — most of their product is sold here and they’re going to be opening up their plants all over the place.”
Are they, though?
It’s true that the branded pharmaceuticals — the household names that left the U.S. for Ireland and its favorable tax regime — need a continued presence in the U.S. because it’s such a big market for pharma, and due to its “relatively short time-to-market for innovative medication,” according to analysts at ING.
“Given their presence all over the globe and their higher margins, some shuffling in their supply chains as a result of tariffs is likely,” they wrote in a briefing.
But will it lead to the full-scale exodus that Trump has promised?
In 2024, pharmaceuticals were the EU’s largest export to the U.S., worth a reported $127 billion.
“You don’t move manufacturing overnight,” said Justine Fassion, an international trade lawyer at Sidley.
“Manufacturing in the pharma industry is subject to various regulatory constraints, so it can take time before you can move production from one country to another,” she said in an interview with POLITICO earlier this year.
Building a manufacturing plant from the ground up generally takes three to five years, much of it related to the local permitting process for utilities, disposal and other community concerns, according to Marta Wosińska, a senior fellow at the Brookings Institute.
“What makes local permits challenging is that they can vary dramatically across locations, increasing the risk for potential delays,” she noted in a briefing.
It’s also extremely costly.
Recent infrastructure expansions in the United States by big-name pharmaceuticals have been led by Eli Lilly’s $23 billion investment into multiple facilities. Merck recently opened a $1 billion plant in North Carolina, where Novo Nordisk is also undertaking a $4.1 billion fill and finish expansion. And Johnson & Johnson is ploughing $2 billion into a biologics facility in New Jersey.
But the cost of site construction might rise further because of the 25 percent tariff Trump has imposed on steel, a major input in industrial construction, she added.
“The same concern applies to manufacturing equipment, which is all stainless steel,” she wrote.
The largest pharma companies warned through their European industry association, EFPIA, that pharma research, development and manufacturing is likely to be redirected toward the U.S., when they sat down for crisis talks with European Commission President Ursula von der Leyen this week.
But the group — conscious that interinstitutional negotiations are about to begin on a Brussels plan to overhaul landmark EU pharma legislation, and the industry’s existing lucrative patent protection with it — added that the migration could be put on hold if Europe “delivers rapid, radical policy change.”
That would mean “strengthening rather than weakening Europe’s intellectual property provisions,” the group said in an emailed statement, as well as “achieving a competitive EU market that attracts, values and rewards innovation in line with other economies at the forefront of patient care.”
Alexander Natz, secretary-general of Eucope, which represents mid-sized pharma companies, said: “Many of our members don’t have factories in the U.S. We can’t just build … within two months, factories in the U.S.”
Smaller and medium-sized pharma companies tend to have one or two innovative products, he said, and they “don’t have a product portfolio to say, ‘well, OK, I can level out the impact of the tariffs on product A, which is produced in that territory, by increasing sales potentially in product B.”
The larger companies that Trump is hoping to entice back might also not think it’s worth the bother.
“These tariffs might not be around forever,” said Jeremy Leonard, managing director of industry services at Oxford Economics.
“It may actually be in your interest to suck up the tariffs and just ride out the storm,” he said, arguing that those companies might instead choose to reduce investments in research and development rather than switch production.
“There certainly are some companies who will do that calculus,” he said. “I don’t think it’s necessarily going to mean a sort of tidal wave of movement, quite frankly.”
What actually encourages companies to move — as Trump alluded to when he called out Dublin — isn’t tariffs, said Ned Hux, a pharmaceutical and life sciences tax partner at PwC.
“Targeted tax incentives, streamlined regulatory approvals, and prioritized government procurement could make U.S.-based production more attractive and competitive,” he said, adding those measures could come in the form of tax deductions, lower tax rates on manufacturing activity, tax credits and low-interest financing for domestic production.
“These approaches offer a proactive way to strengthen U.S. pharmaceutical independence without disrupting global trade relationships,” he added.
And while Trump has criticized Ireland for attracting big pharma companies, he’s also suggested he wants to reshore production of generic drugs from India and China, with an eye to making medicines cheaper for Americans.
ING warns, however, that production in the U.S. would lead to higher prices, as U.S. labor and production costs are higher than those in India, for instance.
Generics lobby Medicines for Europe says that its own analysis of API (active pharmaceutical ingredient) supplier locations for the U.S. market demonstrated that for almost 700 APIs approved in the U.S., Europe is the only supplier.
“The U.S. alone will struggle to build competitive manufacturing and strategic autonomy,” said its director general, Adrian van den Hoven.
“The knock on effect of tariffs will be to drive up costs even higher for American patients.”
"A Section 232 investigation is conducted under the authority of the Trade Expansion Act of 1962, as amended. The purpose of the investigation is to determine the effect of imports on the national security. Investigations may be initiated based on an application from an interested party, a request from the head of any department or agency, or may be self-initiated by the Secretary of Commerce.
The Secretary’s report to the President, prepared within 270 days of initiation, focuses on whether the importation of the article in question is in such quantities or under such circumstances as to threaten to impair the national security. The President can concur or not with the Secretary’s recommendations, and take action to “adjust the imports of an article and its derivatives” or other non-trade related actions as deemed necessary."
https://public-inspection.federalregister.gov/2025-06587.pdf
https://thehill.com/policy/healthcare/5248629-white-house-pharma-semiconductor-tariffs/
White House launches national security investigation into pharma, semiconductors
By Nathaniel Weixel - April 14, 2025The Trump administration disclosed Monday that it had opened an investigation into the effects on national security of importing certain pharmaceuticals, a move widely seen as the prelude to initiating tariffs on prescription drugs.
The Department of Commerce in a Federal Register notice said the probe, known as a Section 232 investigation, began April 1, the day before President Trump announced widespread tariffs on all imported goods but specifically exempted pharmaceuticals.
Section 232 investigations allow the president to restrict imports deemed a threat to national security. Trump last week said he will impose “a major tariff” on prescription drug imports in the coming weeks.
According to the notice, the investigation “includes both finished generic and non-generic drug products, medical countermeasures, critical inputs such as active pharmaceutical ingredients and key starting materials, and derivative products of those items.”
The probe will examine specific issues, including current and projected demand for pharmaceuticals and pharmaceutical ingredients in the U.S.; the role of foreign supply chains, particularly of major exporters, in meeting U.S. demand for pharmaceuticals; the concentration of imports from a small number of suppliers and any associated risks; and the feasibility of increasing domestic capacity for pharmaceuticals and ingredients.
A typical Section 232 investigation takes 270 days before a report is given to the president, but the public comments on the notice are due just three weeks from the official April 16 publication date, an indication the investigation will move much quicker.
Trump launched Section 232 investigations into the copper and timber industries in March. He has already used the investigations to issue tariffs on imported steel, aluminum and cars.
Separately, the Commerce Department said it had launched a similar probe on semiconductor technology. Trump has argued that tariffs on electronic chips will force companies to relocate their factories to the United States.
Fierce Pharma has posted an informative article on the Department of Commerce's Section 232 investigation into pharmaceutical imports:
Trump administration launches national security investigation into pharmaceuticals, paving way for tariffs
By Angus Liu - April 15, 2025 10:40amAn investigation into the national security implications of pharma imports has been underway for two weeks, the Trump administration disclosed Monday.
The Department of Commerce initiated what’s known as a Section 232 investigation targeting pharmaceuticals and related products April 1, according to a Federal Register filing (PDF). A separate probe into semiconductors was launched at the same time.
A Section 232 investigation, enabled by the Trade Expansion Act of 1962, looks into the effects of certain imports on U.S. national security. If a threat is identified, the president can then impose trade restrictions, such as tariffs, to correct the situation or take other non-trade-related actions as deemed necessary, according to an explanation from the Commerce Department.
The new probe targets “both finished generic and non-generic drug products, medical countermeasures, critical inputs such as active pharmaceutical ingredients and key starting materials, and derivative products of those items,” according to the filing.
The Commerce Department will examine topics such as the demand for pharmaceuticals in the U.S., the role of foreign supply chains, the concentration of drug imports from a small number of producers and the potential for foreign countries to weaponize their control over pharma supplies.
After the probe’s scheduled official publication April 16, public comments on the issue will be open for 21 days. An investigation report to President Donald Trump is due in 270 days.
Still, the Trump administration may aim to dramatically beat that deadline. Over the weekend, Commerce Secretary Howard Lutnick said tariffs on the pharma and semiconductor industries will come “in the next month or two.”
Sectoral tariffs on pharmaceuticals, semiconductors and autos are “not available for negotiation” with other countries, Lutnick said Sunday on ABC News.
“They are just going to be part of making sure we reshore the core national security items that need to be made in this country,” he said.
An ongoing 232 investigation doesn’t prevent the Trump administration from imposing tariffs on pharmaceuticals on an emergency basis. Instead, a conclusive Section 232 investigation provides the president justification to add tariffs over longer timelines and to potentially take further actions as he tries to onshore domestic manufacturing. Trump has used findings from previous Section 232 investigations to justify his recent 25% tariffs on steel, aluminum and the auto industry.
In recent months, Trump has threatened 25% or higher tariffs on pharmaceuticals as well. Pharma companies are lobbying the White House for a better deal, including a phase-in approach with a gradual implementation. But, based on the president’s recent comments, industry watchers fear that the tax rate may reach even higher, Jefferies analysts said in a Monday note.
“When the pharmaceutical companies started to go to Ireland, I would have said, 'that’s OK, if you want to go to Ireland. I think that’s great.' But if you want to sell anything into the United States, I’m going to put a 200% tariff on you so you’re never going to be able to sell anything into the United States,” Trump said during a March meeting with Ireland’s leader, Micheál Martin, at the White House.
A 25% tariff would be painful to the pharma industry’s profits, Jefferies analysts concluded. But, overall, the impact is “surmountable for most innovative companies given reasonable gross margins and global manufacturing footprints,” William Blair analysts said in a Monday note.
As the William Blair team sees it, the greatest risk falls on generic drug supplies, which make up 90% of prescriptions in the U.S.
In a statement, the CEO of the Association for Accessible Medicines (AAM), John Murphy, said tariffs will “exacerbate current shortages that hinder patient access” if they are are not joined by “substantive regulatory and reimbursement changes.”
The AAM urges the administration “to work with us on a suite of reforms that prioritize patients, national security, and ultimately will lead to more resilient and reliable access here in the U.S.,” he added.
Get MHF Insights
News and tips for your healthcare freedom.
We never spam you. One-step unsubscribe.