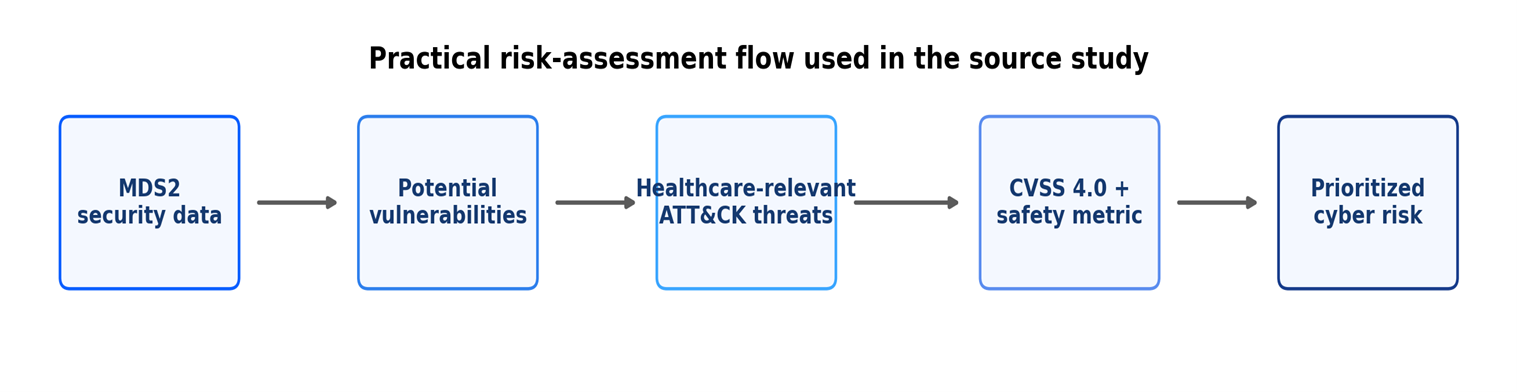
- When your AI agent goes off script: What Moffitt Cancer Center caught early
- Utah physician, 2 nurses charged with $29M fraud scheme
- 3 Ascension CFO moves in 2 days
- The volume paradox costing ASCs millions
- Pediatric radiology leaders launch AI brain tumor dataset
- Inside SCA Health’s clinical nursing ladder — and how it helped cut turnover by 7%
- What ASCs are getting wrong about OR delays — and how to fix it
- 1 behavioral health leader’s playbook for staff retention and safety
- Centene executive to join Froedtert ThedaCare as chief nurse
- Behavioral health ED visits projected to rise by 1 million
- UPMC provides free RN-BSN degree to its nurses
- Physicians aren’t buying payers’ prior auth pledge: 5 survey notes
- 5 dental AI updates in 1 month
- 3 lawsuit settlements in dentistry
- Financial pressures shutter Iowa dental clinics: 4 notes
- 4 cities where systems are winning new grads from tech, finance
- Medline launches predictive supply chain platform and 5 more updates to know
- Independent autism research committee adds 7 members
- FDA Commissioner Marty Makary Resigns After Trump Pressure
- 10 notes on the widening DSO performance gap
- Health systems are betting on retail partnerships
- CDC-linked autism researcher arraigned on fraud charges
- Nearly 1 in 3 boys under 14 discussed suicide in crisis conversations: Report
- How anesthesia became a financial liability for ASCs
- 4 pressures converging on pharmacy
- Ascension Texas hospital taps new assistant CFO
- 82% of physicians are now employed: 6 notes
- Atlanta-area medical office portfolio sold in leaseback deal
- New warning sign for sudden cardiac death found: Study
- NAPA partners with New York hospital
- Remarks at the MFA Legal & Compliance 2026 Conference
- GLP-1 Drugs May Improve Breast Cancer Outcomes
- CMS pauses hospice, home health Medicare enrollments in fraud crackdown
- Merck KGaA looks to M&A to bolster its 'rather slim' pipeline
- Takeda, slimming down for 'new era,' plots 4,500 layoffs in latest restructuring drive
- BeOne Medicines’ Brukinsa TV ad 'Clarity' hit by FDA over 'misleading suggestions'
- Health systems are exploring AI-powered opportunistic screening. New CMS reimbursement could unlock a business case for it
- Most mental health practitioners satisfied with work, financially stable, SimplePractice finds
- Ted Turner's Brain Disease More Common Than Previously Thought, Review Finds
- Novo, Lilly tout respective early response and weight loss maintenance data as GLP-1 rivalry intensifies
- Ivermectin Prescriptions Doubled After Mel Gibson Cancer Cure Claim
- Haleon tackles sports injuries with latest soccer play
- Eating Out Linked To Obesity Risk Worldwide
- Perimenopausal Women Face Greater Heart Risk, Study Finds
- Low Wages, Empty Plates, Heavy Toll: Rethinking Suicide Prevention
- RFK Jr. Swaps Vaccine Talk for Healthy Foods and Reading to Tots in Push To Woo Voters
- Trump and Kennedy Seek To Relax Safeguards for AI Healthcare Tools
- Valneva to lay off up to 15% of workforce in face of ‘adverse trend’ in travel vaccines
- California to award $111M for behavioral health supportive housing
- 6 new psychiatric residency programs to know
- USOSM adds New York practice
- NAMI partners on health crisis preparation hub
- Oklahoma enacts law expanding access to dental care
- Where dentists are leaving value behind in practice sales
- Why dental practices are closing in 2026
- Texas dental school to launch master’s program with orthodontic specialty
- Qualitas Dental Partners makes investments into 7 practices
- Mayo Clinic CEO Gianrico Farrugia stepping down at year's end
- 988 calls are rising — what’s behind the surge?
- Providence hospital to lay off 40 workers amid behavioral health staffing overhaul
- BioMarin consolidates staff at Amicus HQ after closing $4.8B deal for rare disease peer
- US Monitors For Hantavirus As WHO Expects More Cases But 'Not Another COVID'
- University of California, union near May 14 strike deadline with no deal in hand
- 1 in 5 marketplace enrollees dropped their coverage in 2026: media report
- Hims & Hers posts $92M loss in Q1 as it shifts to branded GLP-1 medications
- Listen to the Latest ‘KFF Health News Minute’
- FDA Commissioner Marty Makary to resign, capping turbulent tenure
- FDA Commissioner Marty Makary to resign, capping turbulent tenure
- Providence puts years of losses in rearview with its third consecutive quarter of operating gains
- Millions of Women Suffer in Silence From Treatable Pelvic Organ Prolapse
- Eli Lilly pauses Indian obesity awareness campaign after regulatory notices: report
- Optum Rx unveils new transparent PBM model
- Fitness wearable Whoop adds on-demand clinician access, EHR syncing
- Alkermes’ Lumryz hits phase 3 mark in another sleep disorder, fueling momentum from $2.4B Avadel acquisition
- ACA exchanges take spotlight in Q1
- Pfizer, Arvinas win $85M upfront in Rigel licensing pact for new breast cancer med Veppanu
- Bayer's Eylea declines by 24%, bearing the brunt of biosmilar competition
- As public vaccine criticism quiets, RFK Jr. keeps safety inquiries running in background: NYT
- As public vaccine criticism quiets, RFK Jr. keeps safety inquiries running in background: NYT
- What's Fueling The High U.S. Death Rate? It Might Not Be What You Think
- Telemedicine Not Breaking The Bank, Also Not Expanding Patient Access
- After-School Sports An Overall Boon To Children And Teens, Study Shows
- Trump Promised Cheaper Drugs. Some Prices Dropped. Many Others Shot Up
- Why Are Older Adults Taking Edibles? Survey Reveals Some Surprises
- Low Wages, Empty Plates, Heavy Toll: Rethinking Suicide Prevention
- EU advances scheme to bolster manufacturing autonomy, avert drug shortages
- Bicara Therapeutics hires Replimune, Sanofi alum as chief commercial officer
- The broken pipeline of mental healthcare for LGBTQ teenagers
- FDA Launches One-Day Inspectional Assessments to Strengthen and Expand Oversight
- FDA Launches One-Day Inspectional Assessments to Strengthen and Expand Oversight
- Is your hospital ready for a prolonged IT outage? Joint Commission, AHA's new resiliency program will let you know
- FDA Expands AI Capabilities: Launches ELSA and Completes HALO Data Platform Consolidation
- FDA Expands AI Capabilities: Launches ELSA and Completes HALO Data Platform Consolidation
- Roche acquires PathAI to transform AI-driven diagnostics
- Roche acquires PathAI to transform AI-driven diagnostics
- Trump Planning to Fire FDA Commissioner Marty Makary
- Trump Planning to Fire FDA Commissioner Marty Makary
- Included Health launches AI-powered solution to connect members to providers
- FDA Green Lights Bizengri Drug To Treat Rare, Aggressive Bile Duct Cancer
- The Hidden Design Flaw in Medical Device Service Technology
- The Hidden Design Flaw in Medical Device Service Technology
- An Endovascular Approach to Neurological Diseases Can Shift the Treatment Paradigm
- An Endovascular Approach to Neurological Diseases Can Shift the Treatment Paradigm
- 8,500 Steps A Day Could Be Sweet Spot For Preventing Weight Regain
- Why Gen AI is a Win for MedTech: And, How to Unlock its Potential with the Right Policies
- Why Gen AI is a Win for MedTech: And, How to Unlock its Potential with the Right Policies
- Survey: Employers seeking greater transparency from pharmacy benefits
- Kaiser Permanente's investments pick up the slack as Q1 operating margin slims to 2.1%
- AMA unveils policy framework to combat AI deepfake physician impersonation
- The Medical Device Cybersecurity Gap Hiding in Plain Sight
- The Medical Device Cybersecurity Gap Hiding in Plain Sight
- CSL slashes revenue projection and takes $5B impairment as interim CEO flags R&D misses, market erosion
- Healthcare bankruptcies up 33% in Q1 2026: report
- Why Doctors Are Quitting At An Earlier Age
- Sharper Brains May Face Higher Depression Relapse Risk, Study Finds
- Older Adults Have Fewer Regrets, Study Says
- Partner's bispecific Bizengri nabs FDA national priority nod in rare bile duct cancer
- Daiichi Sankyo targets global top 5 oncology rank by 2035, $1.3B efficiency drive in new 5-year plan
- That Discount At The Pharmacy Counter May Pack Hidden Costs
- Nighttime Heat Waves Increase Asthma Risk
- Watch: 8 Health Insurance Terms You Should Know
- As Ranks of Uninsured Grow, Minnesota’s Hospitals Are Among Least Charitable in Nation
- OVID Health hires Edelman alum Davide Scalenghe to boost its international footprint
- Maintaining trust in medical AI: Monitoring and managing model lifecycle
- Maintaining trust in medical AI: Monitoring and managing model lifecycle
- Eli Lilly shoots for health in new Caitlin Clark ad campaign
- Omada Health posts 42% revenue jump in Q1, joins Eli Lilly employer weight loss program
- Journalists Shed Light on Deadly Hantavirus Outbreak and a Crisis in the Nation’s ERs
- The Make America Healthy Again Movement Comes for Hospital Food
- Remarks at the Conference on Financial Market Regulation
- Dad Jokes: Remarks at the 13th Annual Conference on Financial Markets Regulation
- RFK Jr. Launches Plan To Curb Antidepressant 'Overprescription'
- Skil-Care launches specialized healthcare product innovation program
- Sanford Health unveils deal to integrate Minnesota-area North Memorial Health, invest $600M
- UPDATED—Trump plans to fire FDA chief Marty Makary: report
- Remarks at the Special Competitive Studies Project AI+ Expo
- Plant-Based Foods May Help Lower Risk of High Blood Pressure
- Integrated CDO capabilities reduce early development complexity
- Targeted Protein Degradation and Novel Modalities: Getting on the Frontline
- Workplace safety is a top priority for 93% of healthcare leaders: Axon survey
- Super Shoes Might Increase Risk Of Running Injuries, Study Says
- TV, Movies Offer Flawed Depictions Of Autism, Add To Delayed Diagnosis, Study Says
- Opioid OD Survivors Have Triple Rate Of Repeat Overdoses Than Previously Estimated
- A New Medicare Option For Weight Loss Drugs: What Older Americans Should Know
- Exposure Therapy Can Successfully Ease Peanut Allergies
- Listen: A Federal Agency Is After Workers’ Health Data, and Critics Are Alarmed
- In California Governor Race, Single-Payer Is a Litmus Test. There’s Still No Way To Pay for It.
- Cruise Ship Hantavirus Outbreak Kills 3 as WHO Says Risk Is Low
- Remarks at the 13th Annual Conference on Financial Market Regulation
- New Study Suggests The Brain Can Continue Learning While In An Unconscious State

Statement of the American Hospital Association for the Committee on Energy and Commerce of the U.S. House of Representatives
May 24, 2023
On behalf of our nearly 5,000 member hospitals, health systems and other health care organizations, our clinician partners — including more than 270,000 affiliated physicians, 2 million nurses and other caregivers — and the 43,000 health care leaders who belong to our professional membership groups, the American Hospital Association (AHA) writes to share the hospital field’s comments on legislative proposals that are to be considered before the Energy and Commerce Committee on May 24.
We would like to provide feedback on sections of H.R. 3561 as amended (HEALTHMARK-FC-AINS_01.XML), as well as H.R. 3290 as amended (H3290-FC-AINS_01.XML).
MEDICAID DSH LEGISATION
Delaying Certain Disproportionate Share Hospital Payment Reductions under the Medicaid Program
The AHA supports Section 203 to address the Medicaid disproportionate share hospital (DSH) reductions. Congress established the Medicaid DSH program to provide financial assistance to hospitals serving a disproportionate number of low-income patients to ensure Medicaid and uninsured patients have access to health care services. These hospitals also provide critical community services, such as trauma and burn care, maternal and child health care, high-risk neonatal care and disaster preparedness resources. The patients they serve are among those that need care the most and often experience challenges accessing it, including children, the poor, the disabled and the elderly.
Reductions to the Medicaid DSH program were enacted as part of the Affordable Care Act, with the reasoning that hospitals would have less uncompensated care as health insurance coverage increased. Under current statute, the Medicaid DSH cut is $8 billion for each of the next four fiscal years (2024-2027, for a total of $32 billion for all four years). Unfortunately, the projected coverage levels have not been realized and hospitals continue to care for patients for whom they are not receiving payment. Consequently, the need for Medicaid DSH payments is still vital for the hospitals that rely on the program.
PRICE TRANSPARENCY LEGISLATION
Price Transparency Requirements
We appreciate the Committee working to modify Section 101 that is focused on hospital price transparency and we look forward to continuing to engage on these efforts. The hospital field has shown its commitment to adhering to the Hospital Price Transparency Rule. The Centers for Medicare & Medicaid Services (CMS) found that in 2022, 70% of hospitals complied with both components of the Hospital Price Transparency Rule, including the consumer-friendly display of shoppable services information, as well as the machine-readable file requirements. This is an increase from 27% in 2021. Moreover, when looking at each individual component of the rule, 82% of hospitals met the consumer-friendly display of shoppable services information requirement in 2022 (up from 66% in 2021) and 82% met the machine-readable file requirement (up from 30% in 2021).
MEDICARE SITE NEUTRAL LEGISATIVE PROPOSALS
Parity in Medicare Payments for Hospital Outpatient Department Services Furnished Off-Campus
The AHA opposes Section 302. This provision would create additional site-neutral payment cuts over four years for the administration of drug services furnished in an off-campus provider-based department. We are especially concerned that this would result in a major cut for hospital outpatient departments (HOPDs) that provide essential drug administration services, including for vulnerable cancer patients, who may require a higher level of care as they receive their essential treatments. According to AHA data, this proposal would result in a cut to hospitals of $54.2 million in the first year and $3 billion over 10 years. More information about the impact of this proposal at the state level, as well as the national and state impacts of the two additional site-neutral legislative proposals that were considered by the Health Subcommittee on April 26, can be found here.
This legislative effort would expand existing site-neutral payment cuts, which have already had a significantly negative impact on the financial sustainability of hospitals and health systems and have contributed to Medicare’s chronic failure to cover the cost of caring for its beneficiaries. According to the Medicare Payment Advisory Commission (MedPAC), overall Medicare hospital margins were negative 6.3% in 2021 after accounting for temporary COVID-19 relief funds. Without these funds, the overall Medicare margin for 2021 remained depressed at negative 8.2% after hitting a staggering low of negative 12.3% in 2020. On average, Medicare only pays 84 cents for every dollar hospitals spend providing care to Medicare beneficiaries. Moreover, overall median hospital operating margins were negative throughout 2022 and into the beginning of 2023. Site-neutral cuts have already contributed to these shortfalls and any further expansion of these policies will exacerbate this situation and threaten patients’ access to quality care.
This legislation also fails to account for the fundamental differences between HOPDs and other sites of care. The cost of care delivered in hospitals and health systems takes into account the unique benefits that they provide to their communities. This includes the investments made to maintain standby capacity for natural and man-made disasters, public health emergencies and unexpected traumatic events, as well as deliver 24/7 emergency care to all who come to the hospital, regardless of ability to pay or insurance status. This standby role is built into the cost structure of hospitals and is supported by revenue from direct patient care — a situation that does not exist for any other type of provider. Expanding site-neutral cuts to HOPDs and the outpatient services they provide would endanger the critical role they play in their communities, including access to care for patients.
Additionally, hospital facilities treat patients who are sicker and have more chronic conditions than those treated in physician offices or ambulatory surgical centers. Hospitals are better equipped to handle complications and emergencies, but this often requires the use of additional resources that other settings do not typically provide. Hospital facilities also must comply with a much more comprehensive scope of licensing, accreditation and other regulatory requirements compared to other sites of care.
Requiring a Separate Identification Number and an Attestation for Each Off-Campus Outpatient Department of a Provider
The AHA opposes Section 103 that requires that each outpatient department of a provider be assigned a separate unique health identifier from its provider. The Department of Health and Human Services (HHS) Secretary would be required to issue rulemaking to establish a process to review each separate unique health identifier assigned to each outpatient department of a provider and confirm the provider is compliant with the provider-based requirements in the Social Security Act. This also would require that hospitals with outpatient departments submit an attestation of compliance with the provider-based regulations for each of their outpatient departments as a condition of payment.
We are very concerned about this requirement given that past CMS review and approval of similar attestations has been extremely burdensome and difficult. For example, CMS contractors did not complete their audits for the “mid-build” exception to the current site-neutral payment rates until over two years after the statutory deadline. In addition, it soon became clear that these audits were conducted inaccurately — and so incorrectly that CMS itself rescinded all audit denials and re-reviewed all exception applications.
HEALTH-RELATED OWNERSHIP INFORMATION
Mandatory Reporting with Respect to Certain Health-Related Ownership Information
The AHA opposes Section 104. The AHA has serious concerns about this provision. It is overly burdensome — for example, it would require certain hospitals to report every time a physician chooses to take a job elsewhere. It also is redundant to other reporting. CMS already requires reporting on private equity transactions and the Federal Trade Commission and the Department of Justice require information regarding large mergers and acquisitions before they occur through antitrust Hart, Scott, Rodino Act reporting. In addition, the HHS Secretary would have unrestricted discretion to add other reporting requirements, which will lead to additional burdens for hospitals and health systems.
PHARMACY BENEFIT MANAGER LEGISLATION
Improving Transparency and Preventing the Use of Abusive Spread Pricing and Related Practices in Medicaid
The AHA is concerned about language included in Section 303 that would require 340B entities to report the difference between their acquisition cost and payments from Medicaid managed care organizations (MCOs). Such reporting would not only overstate how much 340B hospitals save from the program for their Medicaid beneficiaries but would also be unnecessarily burdensome and costly to 340B entities. We do appreciate the Committee amending this section to allow Medicaid MCOs to pay 340B entities above acquisition cost to continue to ensure 340B hospitals can maintain access to these critical savings for Medicaid MCO patients. However, the addition of burdensome reporting requirements is problematic, and we urge the Committee to strike this language.
340B TRANSPARENCY ACT
The AHA opposes H.R. 3290, the 340B Transparency Act. While we appreciate the Committee’s longstanding support for the 340B program and interest in increasing program transparency, H.R. 3290 would impose onerous transparency requirements that will not provide policymakers with meaningful information on the true value of the program to providers and their patients. The proposal would impose new and burdensome reporting requirements that would require hospitals to implement a number of new programs and systems to collect data at each outpatient location where 340B drugs may be dispensed or administered. Specifically, the proposal would require 340B hospitals to report by location the total number of individuals receiving 340B drugs by payer, total costs, payments and savings. None of these data points individually or collectively will tell the full story of how 340B hospitals use the program to benefit the patients and communities they serve. In addition, the proposal would grant the HHS Secretary authority to impose civil monetary penalties on 340B hospitals for failure to comply with the new reporting requirements and allow the HHS Secretary the discretion to determine the penalty amount.
For example, charity care numbers only indicate the amount of care provided to patients who qualify for the hospital’s financial assistance policy and is therefore provided to the patient free of or below cost. It does not account for the costs that hospitals incur in caring for underinsured patients or patients covered by government programs that chronically underpay the cost of care or the many other programs and services 340B savings support. Looking at charity care would obscure the breadth of programs and services that are most often supported by 340B savings, such as free clinics, behavioral health treatment, trauma care and diabetes counseling programs.
Requiring these measures to be reported at each outpatient location is overly burdensome and will not reflect the value 340B provides to the patients and communities eligible hospitals serve. Savings from each location are used to establish and support a variety of programs that benefit entire communities. In addition, hospitals will be forced to incur additional costs to be able to report at the level of detail required — costs that will be difficult to bear for many hospitals that are already facing myriad financial challenges. Over half of hospitals in the country ended 2022 operating in the red, in large part due to double-digit increases in expenses from labor, drugs, as well as supplies and equipment. Imposing additional costs on hospitals and the threat of penalty for failure to report would be egregious and unsustainable for the field.
Furthermore, the bill would not only add unnecessary cost and burden but also fail to consider the fact that 340B hospitals already report a number of data on how much they invest in resources to benefit the community. 340B hospitals must annually recertify their eligibility to participate and attest to meeting all the program requirements; participate in audits conducted by the Health Resources and Services Administration (HRSA) and drug manufacturers; and maintain auditable records and inventories of all 340B and non-340B prescription drugs. In addition, 340B hospitals report data on the benefits they are providing to their communities through the IRS tax form 990 submitted by all nonprofit hospitals. These data show that in 2019 alone, 340B hospitals provided nearly $68 billion in community benefits. Finally, 340B hospitals also already report their total charity care costs as well as their uncompensated and unreimbursed care costs on the Medicare cost reports filed annually with CMS.
Instead, the AHA encourages the Committee to address the alarming efforts by many of the largest drug companies to restrict or outright deny access to 340B discounted drugs through lawful arrangements 340B hospitals have established with community and specialty pharmacies to improve access to care. Since July 2020, over 20 drug companies have adopted these restrictive policies to the severe detriment of 340B hospitals and their patients. The financial impact on 340B hospitals is significant. For 340B critical access hospitals, AHA’s survey reports average annual losses of over $500,000 and 340B disproportionate share hospitals have average annual losses of nearly $3 million. These impacts have meant reduced access to the many programs and services that patients rely on for care, and for some rural hospitals, has put their financial health in jeopardy. The AHA continues to actively support HRSA in its efforts to enforce the law and hold drug companies accountable. The 340B program is a lifeline for hospitals across the country, no more evident than during the pandemic when hospitals and their staff served on the frontlines, and such actions by drug companies threaten the program’s viability for providers and their patients.
CONCLUSION
Thank you for your consideration of the AHA’s comments on these legislative proposals. We look forward to continuing to work with you to address these important topics on behalf of our patients and communities.
Get MHF Insights
News and tips for your healthcare freedom.
We never spam you. One-step unsubscribe.